




Tumors can carry mutations in hundreds of different genes, and each of those genes may be mutated in different ways — some mutations simply replace one DNA nucleotide with another, while others insert or delete larger sections of DNA.
Until now, there has been no way to quickly and easily screen each of those mutations in their natural setting to see what role they may play in the development, progression, and treatment response of a tumor. Using a variant of CRISPR genome-editing known as prime editing, MIT researchers have now come up with a way to screen those mutations much more easily.
The researchers demonstrated their technique by screening cells with more than 1,000 different mutations of the tumor suppressor gene p53, all of which have been seen in cancer patients. This method, which is easier and faster than any existing approach, and edits the genome rather than introducing an artificial version of the mutant gene, revealed that some p53 mutations are more harmful than previously thought.
This technique could also be applied to many other cancer genes, the researchers say, and could eventually be used for precision medicine, to determine how an individual patient’s tumor will respond to a particular treatment.
“In one experiment, you can generate thousands of genotypes that are seen in cancer patients, and immediately test whether one or more of those genotypes are sensitive or resistant to any type of therapy that you’re interested in using,” says Francisco Sanchez-Rivera, an MIT assistant professor of biology, a member of the Koch Institute for Integrative Cancer Research, and the senior author of the study.
MIT graduate student Samuel Gould is the lead author of the paper, which appears today in Nature Biotechnology.
Editing cells
The new technique builds on research that Sanchez-Rivera began 10 years ago as an MIT graduate student. At that time, working with Tyler Jacks, the David H. Koch Professor of Biology, and then-postdoc Thales Papagiannakopoulos, Sanchez-Rivera developed a way to use CRISPR genome-editing to introduce into mice genetic mutations linked to lung cancer.
In that study, the researchers showed that they could delete genes that are often lost in lung tumor cells, and the resulting tumors were similar to naturally arising tumors with those mutations. However, this technique did not allow for the creation of point mutations (substitutions of one nucleotide for another) or insertions.
“While some cancer patients have deletions in certain genes, the vast majority of mutations that cancer patients have in their tumors also include point mutations or small insertions,” Sanchez-Rivera says.
Since then, David Liu, a professor in the Harvard University Department of Chemistry and Chemical Biology and a core institute member of the Broad Institute, has developed new CRISPR-based genome editing technologies that can generate additional types of mutations more easily. With base editing, developed in 2016, researchers can engineer point mutations, but not all possible point mutations. In 2019, Liu, who is also an author of the Nature Biotechnology study, developed a technique called prime editing, which enables any kind of point mutation to be introduced, as well as insertions and deletions.
“Prime editing in theory solves one of the major challenges with earlier forms of CRISPR-based editing, which is that it allows you to engineer virtually any type of mutation,” Sanchez-Rivera says.
When they began working on this project, Sanchez-Rivera and Gould calculated that if performed successfully, prime editing could be used to generate more than 99 percent of all small mutations seen in cancer patients.
However, to achieve that, they needed to find a way to optimize the editing efficiency of the CRISPR-based system. The prime editing guide RNAs (pegRNAs) used to direct CRISPR enzymes to cut the genome in certain spots have varying levels of efficiency, which leads to “noise” in the data from pegRNAs that simply aren’t generating the correct target mutation. The MIT team devised a way to reduce that noise by using synthetic target sites to help them calculate how efficiently each guide RNA that they tested was working.
“We can design multiple prime-editing guide RNAs with different design properties, and then we get an empirical measurement of how efficient each of those pegRNAs is. It tells us what percentage of the time each pegRNA is actually introducing the correct edit,” Gould says.
Analyzing mutations
The researchers demonstrated their technique using p53, a gene that is mutated in more than half of all cancer patients. From a dataset that includes sequencing information from more than 40,000 patients, the researchers identified more than 1,000 different mutations that can occur in p53.
“We wanted to focus on p53 because it’s the most commonly mutated gene in human cancers, but only the most frequent variants in p53 have really been deeply studied. There are many variants in p53 that remain understudied,” Gould says.
Using their new method, the researchers introduced p53 mutations in human lung adenocarcinoma cells, then measured the survival rates of these cells, allowing them to determine each mutation’s effect on cell fitness.
Among their findings, they showed that some p53 mutations promoted cell growth more than had been previously thought. These mutations, which prevent the p53 protein from forming a tetramer — an assembly of four p53 proteins — had been studied before, using a technique that involves inserting artificial copies of a mutated p53 gene into a cell.
Those studies found that these mutations did not confer any survival advantage to cancer cells. However, when the MIT team introduced those same mutations using the new prime editing technique, they found that the mutation prevented the tetramer from forming, allowing the cells to survive. Based on the studies done using overexpression of artificial p53 DNA, those mutations would have been classified as benign, while the new work shows that under more natural circumstances, they are not.
“This is a case where you could only observe these variant-induced phenotypes if you’re engineering the variants in their natural context and not with these more artificial systems,” Gould says. “This is just one example, but it speaks to a broader principle that we’re going to be able to access novel biology using these new genome-editing technologies.”
Because it is difficult to reactivate tumor suppressor genes, there are few drugs that target p53, but the researchers now plan to investigate mutations found in other cancer-linked genes, in hopes of discovering potential cancer therapies that could target those mutations. They also hope that the technique could one day enable personalized approaches to treating tumors.
“With the advent of sequencing technologies in the clinic, we’ll be able to use this genetic information to tailor therapies for patients suffering from tumors that have a defined genetic makeup,” Sanchez-Rivera says. “This approach based on prime editing has the potential to change everything.”
The research was funded, in part, by the National Institute of General Medical Sciences, an MIT School of Science Fellowship in Cancer Research, a Howard Hughes Medical Institute Hanna Gray Fellowship, the V Foundation for Cancer Research, a National Cancer Institute Cancer Center Support Grant, the Ludwig Center at MIT, a Koch Institute Frontier Award, the MIT Research Support Committee, and the Koch Institute Support (core) Grant from the National Cancer Institute.
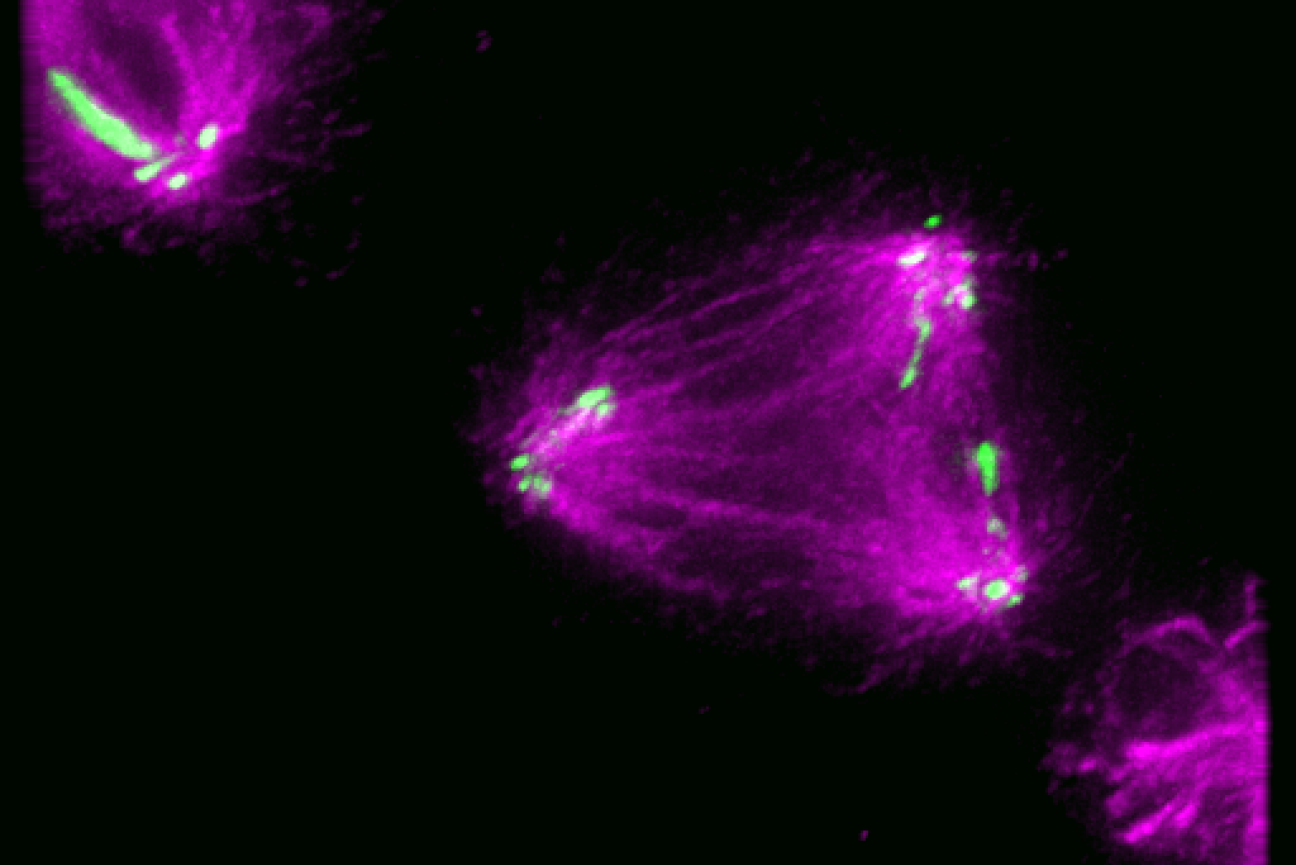
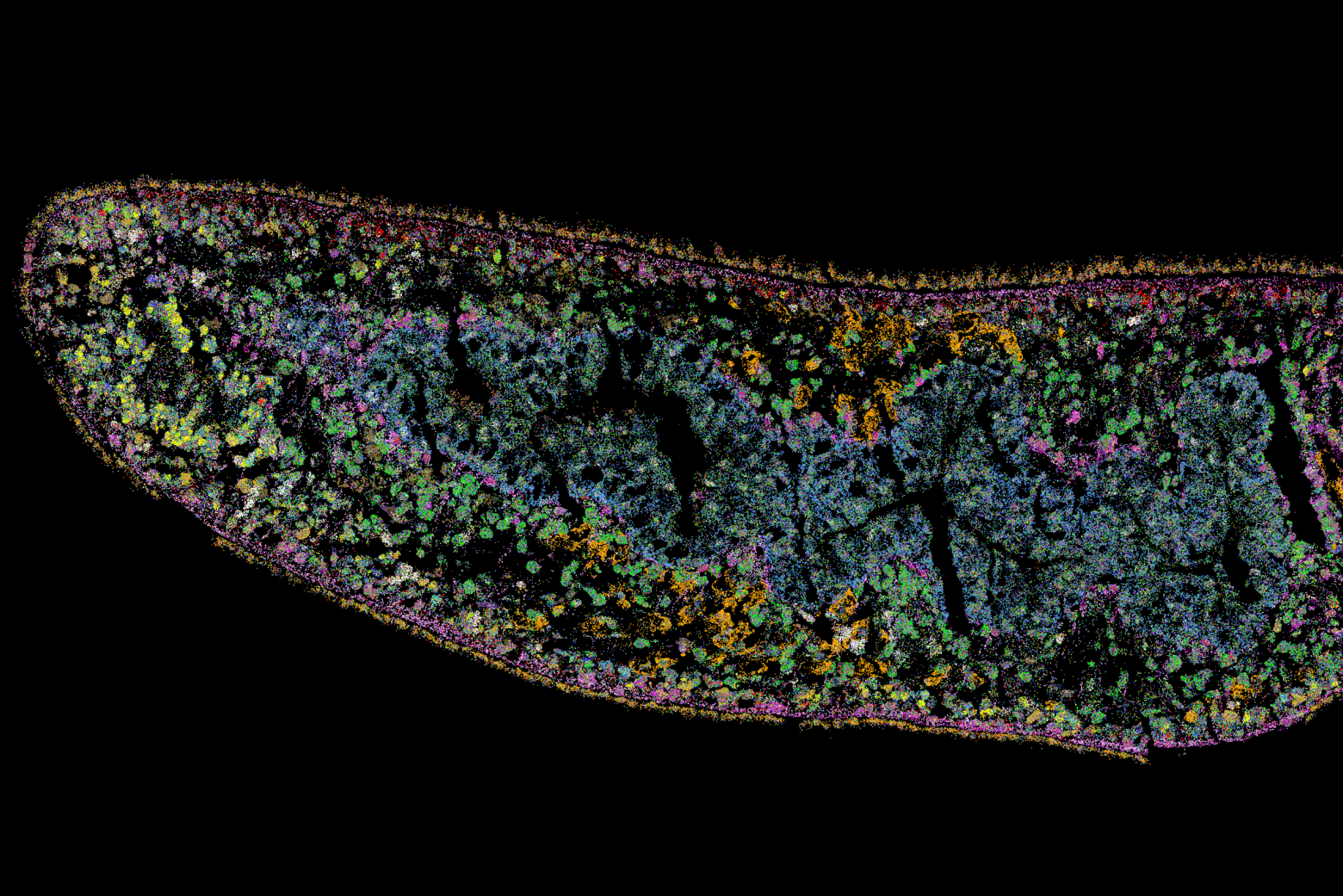
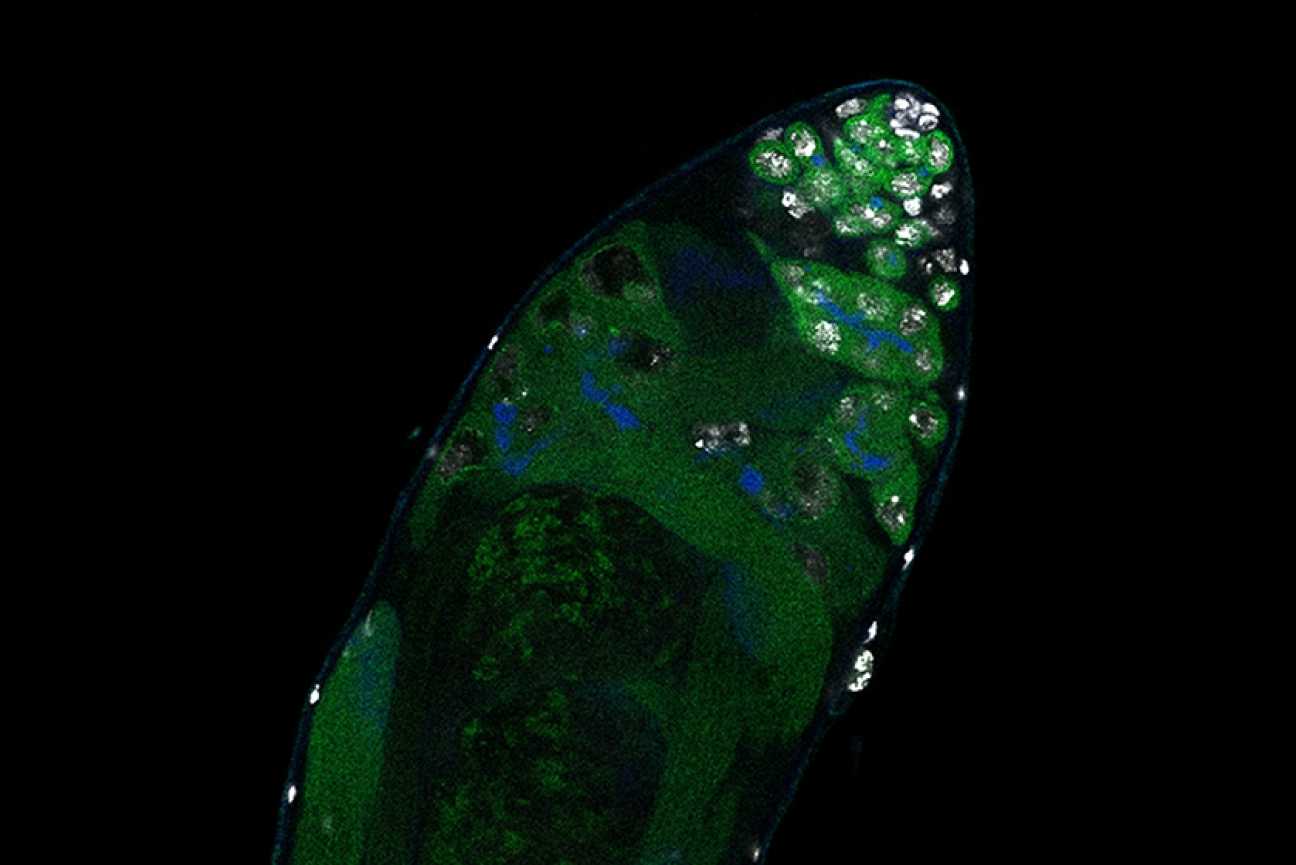
Germline stem cells are the pool of stem cells capable of becoming eggs or sperm. They divide asymmetrically, such that one of the cells resulting from a division is another stem cell and the other is a differentiated cell, which has progressed one step further down the path towards becoming an egg or sperm. Researchers have thought that this asymmetrical division served to replenish the pool of stem cells—making sperm or eggs, but also making more stem cells to produce future sperm or eggs. However, the germline has another way to replenish itself: cells that have differentiated only one or two steps down the path to becoming eggs or sperm are capable of reverting into stem cells. Why, then, do stem cells divide asymmetrically?
New research from Whitehead Institute Member Yukiko Yamashita, who is also a professor of biology at the Massachusetts Institute of Technology and an HHMI Investigator, and former postdoc in her lab Jonathan Nelson shows that asymmetrical division in germline stem cells serves a different but equally important purpose in male fruit flies (Drosophila melanogaster), a common model animal for germline research. The work, published in the journal Proceedings of the National Academy of Sciences (PNAS) on November 13, suggests that in flies, germline stem cells divide asymmetrically in order to unequally split a certain kind of DNA, called ribosomal DNA (rDNA), between the two dividing cells and then keep the cell with more rDNA in the stem cell pool. This is necessary in order to keep the germline viable over generations of cell divisions, and so to keep individual flies fertile and capable of reproduction. The researchers show that only germline stem cells, and not other types of germ cells, drive this process, and explain why stem cells’ asymmetric divisions make them uniquely suited to maintaining rDNA.
Ribosomal DNA is critical to maintain in the germline because it contains the instructions for making a major part of ribosomes, the cellular machines that build proteins from genetic instructions. Proteins are the main workhorses of the cell, and so cells need to make many ribosomes in order to build all of the proteins that they need. Consequently, rDNA exists as many copies repeated in a row of the code for components of the ribosome. All of these repeats make it easy for the cell to mass produce ribosomes, but they also come with a risk: repetitive DNA is prone to losing repeats during cell division. When the cell’s rDNA is copied, it’s easy for a few of the many identical repeats to get cut out, so that the resulting copy of the genome has fewer rDNA repeats than the original.
Most cells can afford to lose a few rDNA repeats without too many negative effects, but the germline cannot. Whereas other cells die with the body they are in, germ cells produce eggs and sperm that will form a new body, which produces new germ cells, and so on. The germ cell lineage is effectively immortal. Over the course of its endless cycle of cell division, the loss of rDNA repeats would add up until the cells became dysfunctional and then died. This would make the individual bearing those germ cells infertile, and so cause their lineage to go extinct.
Researchers have known that germ cells have some way to regain rDNA repeats when the number gets too low—if germ cells couldn’t do this, none of us would exist—but the details of how cells achieve this have been largely mysterious. One proposed model was that when a germ cell divides, sometimes it might divide up its rDNA unequally between the two resulting cells, so that one cell would gain rDNA repeats. Yamashita and Nelson have previously found evidence that this model is correct, and they discovered some of the specific mechanisms that enable it to happen. In a 2023 PNAS paper, the researchers showed that a retrotransposon, a “selfish” genetic element whose function is to make more copies of itself, actually helps germ cells maintain rDNA. During cell division, the retrotransposon R2 slices open one copy of the chromosome containing rDNA in its quest to insert extra copies of itself into the genome. The cell tries to repair the break using the copy on the other intact chromosome, but the tricky nature of repetitive DNA can cause the cell to lose its place, so that it stitches a stretch of rDNA repeats from one copy of the chromosome into the other copy instead.
Through this process, the germline can boost the level of rDNA in a cell—but only by as much as another cell loses. How does this win-lose exchange lead to an overall increase in rDNA levels across the germline cell population to compensate for lost rDNA? In this latest work, Yamashita and Nelson show through mathematical modeling that in cells that divide symmetrically, it would not. Gains and losses in rDNA through this form of exchange would occur essentially at random and cancel each other out over time.
Now consider an asymmetric division. After a germline stem cell divides, the cell that differentiates will go through a few more divisions and ultimately create a specific number of sperm cells–the number happens to be sixty-four. If this daughter cell gets the chromosome with more rDNA repeats, then that would lead to sixty-four sperm with more rDNA repeats—but that would be it, as the sperm have exited the pool of replicating germline stem cells.
However, the daughter cell that remains a germline stem cell will divide again to create a differentiated cell (which will become sixty-four sperm) and another stem cell, which will divide again, leading to another sixty-four sperm and another stem cell—and so on. All of these cells, including many sperm, would inherit the higher number of rDNA repeats. Furthermore, at each division, there would be an opportunity for another unequal split of rDNA. As long as the stem cell always gets the boost in rDNA, then the cumulative number of rDNA repeats would keep growing in the overall population over time—and Yamashita and colleagues’ past work shows that the germline can ensure this. A 2022 Science Advances paper from Yamashita and then-postdoc in her lab George Watase showed that when a germline stem cell divides, the DNA strand with more rDNA repeats is tagged with a protein that the researchers named Indra, which helps mark it to stay in the daughter cell that will become another stem cell. Yamashita and Nelson’s new paper includes mathematical modeling by second author Tomohiro Kumon, a postdoc in Yamashita’s lab, that proves that this is not only sufficient to restore the level of rDNA repeats over time, but that it is the most effective and efficient way for the germline to do so.
“There was this problem with the unequal exchange model of rescuing rDNA, because every cell that gained rDNA did so at the expense of another that was losing it,” Nelson says. “What we show here is that the reason why there’s a bias towards gain in the germline is because this process is happening within these asymmetrically dividing germline stem cells that can gain and gain and gain, while the cells that lose rDNA exit the cycle and so have a limited effect.”
The researchers complemented their mathematical modeling with evidence that the process to increase rDNA repeats occurs primarily or solely in germline stem cells. They found that when the number of rDNA repeats got low enough, then expression of R2 and the presence of double-stranded DNA breaks both increased in germline stem cells, but not significantly in other germ cell types.
Yamashita and Nelson propose that the different cell types in the germline take on different functions to create a pipeline for maximizing the health of future sperm. Germ cells that are one or two steps down the path of differentiation from stem cells are essentially identical to them, to the point that they can be difficult to tell apart in testing, but they divide symmetrically. They are also much more sensitive to DNA damage; the researchers found that R2 exposure kills these cells.
Germline stem cells, with their asymmetrical division and ability to tolerate R2 expression, serve to restore rDNA levels when they get too low. Then the differentiated germ cells serve to weed out mutations—including those introduced during R2 expression in the earlier stem cell stage—by killing off cells with DNA damage. The different strengths of the different types of germ cells creates an effective pipeline to produce the largest number of sperm cells with high rDNA repeat number and low DNA damage.
Eventually, this new understanding of the details of how cells maintain their rDNA could lead to medical therapies. For example, cancer cells are, like germ cells, an essentially immortal cell line, and so must have a way to maintain their rDNA. If researchers could someday find a way to prevent them from doing so, that could be a good treatment strategy. The work also may have implications for research on aging, as rDNA decreases with age in other cell types. In the meantime, Yamashita and Nelson are excited to have solved several long-standing mysteries in their field, including how germ cells can restore rDNA at a population level when each division creates an equal loss and gain of rDNA, and why germline stem cells divide asymmetrically.
“Typically, when you publish a paper, you feel like you’ve fit two puzzle pieces together, but in this case, I feel like we fit a bunch of puzzle pieces together,” Yamashita says. “It’s been immensely satisfying to find answers to multiple questions and see how they all fit together to explain the mechanisms of this process that’s necessary for germline immortality.”

Sipping a beer on a warm summer evening, one might not consider that humans and yeast have been inextricably linked for thousands of years; winemaking, baking, and brewing all depend on budding yeast. Outside of baking and fermentation, researchers also use Saccharomyces cerevisiae, classified as a fungus, to study fundamental questions of cell biology.
Budding yeast gets its name from the way it multiplies. A daughter cell forms first as a swelling, protruding growth on the mother cell. The daughter cell projects further and further from the mother cell until it detaches as an independent yeast cell.
How do cells decide on a front and back? How do cells decode concentration gradients of chemical signals to orient in useful directions, or sense and navigate around physical obstacles? New Department of Biology faculty member Daniel “Danny” Lew uses the model yeast S. cerevisiae, and a non-model yeast with an unusual pattern of cell division, to explore these questions.
Q: Why is it useful to study yeast, and how do you approach the questions you hope to answer?
A: Humans and yeast are descended from a common ancestor, and some molecular mechanisms developed by that ancestor have been around for so long that yeast and mammals often use the same mechanisms. Many cells develop a front and migrate or grow in a particular direction, like the axons in our nervous system, using similar molecular mechanisms to those of yeast cells orienting growth towards the bud.
When I started my lab, I was working on cell cycle control, but I’ve always been interested in morphogenesis and the cell biology of how cells change shape and decide to do different things with different parts of themselves. Those mechanisms turn out to be conserved between yeast and humans.
But some things are very different about fungal and animal cells. One of the differences is the cell wall and what fungal cells have to do to deal with the fact that they have a cell wall.
Fungi are inflated by turgor pressure, which pushes their membranes against the rigid cell wall. This means they’ll die if there is any hole in the cell wall, which would be expected to happen often as cells remodel the wall in order to grow. We’re interested in understanding how fungi sense when any weak spots appear in the wall and repair them before those weak spots become dangerous.
Yeast cells, like most fungi, also mate by fusing with a partner. To succeed, they must do the most dangerous thing in the fungal lifecycle: get rid of the cell wall at the point of contact to allow fusion. That means they must be precise about where and when they remove the wall. We’re fascinated to understand how they know it is safe to remove the wall there, and nowhere else.
We take an interdisciplinary approach. We’ve used genetics, biochemistry, cell biology, and computational biology to try and solve problems in the past. There’s a natural progression: observation and genetic approaches tend to be the first line of attack when you know nothing about how something works. As you learn more, you need biochemical approaches and, eventually, computational approaches to understand exactly what mechanism you’re looking at.
I’m also passionate about mentoring, and I love working with trainees and getting them fascinated by the same problems that fascinate me. I’m looking to work with curious trainees who love addressing fundamental problems.
Q: How does yeast decide to orient a certain way—towards a mating partner, for example?
A: We are still working on questions of how cells analyze the surrounding environment to pick a direction. Yeast cells have receptors that sense pheromones that a mating partner releases. What is amazing about that is that these cells are incredibly small, and pheromones are released by several potential partners in the neighborhood. That means yeast cells must interpret a very confusing landscape of pheromone concentrations. It’s not apparent how they manage to orient accurately toward a single partner.
That got me interested in related questions. Suppose the cell is oriented toward something that isn’t a mating partner. The cell seems to recognize that there’s an obstacle in the way, and it can change direction to go around that obstacle. This is how fungi get so good at growing into things that look very solid, like wood, and some fungi can even penetrate Kevlar vests.
If they recognize an obstacle, they have to change directions and go around it. If they recognize a mating partner, they have to stick with that direction and allow the cell wall to get degraded. How do they know they’ve hit an obstacle? How do they know a mating partner is different from an obstacle? These are the questions we’d like to understand.
Q: For the last couple of years, you’ve also been studying a budding yeast that forms multiple buds when it reproduces instead of just one. How did you come across it, and what questions are you hoping to explore?
A: I spent several years trying to figure out why most yeasts make one bud and only one bud, which I think is related to the question of why migrating cells make one and only one front. We had what we thought was a persuasive answer to that, so seeing a yeast completely disobey that and make as many buds as it felt like was a shock, which got me intrigued.
We started working on it because my colleague,Amy Gladfelter, had sampled the waters around Woods Hole, Massachusetts. When she saw this specimen under a microscope, she immediately called me and said, “You have to look at this.”
A question we’re very intrigued by is if the cell makes five, seven, or 12 buds simultaneously, how do they divide the mother cell’s material and growth capacity five, seven, or 12 ways? It looks like all of the buds grow at the same rate and reach about the same size. One of our short-term goals is to check whether all the buds really get to exactly the same size or whether they are born unequal.
And we’re interested in more than just growth rate. What about organelles? Do you give each bud the same number of mitochondria, nuclei, peroxisomes, and vacuoles? That question will inevitably lead to follow-up questions. If each bud has the same number of mitochondria, how does the cell measure mitochondrial inheritance to do that? If they don’t have the same amount, then buds are each born with a different complement and ratio of organelles. What happens to buds if they have very different numbers of organelles?
As far as we can tell, every bud gets at least one nucleus. How the cell ensures that each bud gets a nucleus is a question we’d also very much like to understand.
We have molecular candidates because we know a lot about how model yeasts deliver nuclei, organelles, and growth materials from the mother to the single bud. We can mutate candidate genes and see if similar molecular pathways are involved in the multi-budding yeast and, if so, how they are working.
It turns out that this unconventional yeast has yet to be studied from the point of view of basic cell biology. The other thing that intrigues me is that it’s a poly-extremophile. This yeast can survive under many rather harsh conditions: it’s been isolated in Antarctica, from jet engines, from all kinds of plants, and of course from the ocean as well. An advantage of working with something so ubiquitous is we already know it’s not toxic to us under almost any circumstances. We come into contact with it all the time. If we learn enough about its cell biology to begin to manipulate it, then there are many potential applications, from human health to agriculture.