When Hanjun Lee arrived at MIT, he was set on becoming a Course 5 chemistry student. Based on his experience in high school, biology was all about rote memorization.
That changed when he took course 7.03 (Genetics), taught by then-professor Aviv Regev, now head and executive vice president of research and early development at Genentech, and Peter Reddien, professor of biology and core member and associate director of the Whitehead Institute for Biomedical Research.
He notes that friends from other schools don’t cite a single course that changed their major, but he’s not alone in choosing Course 7 because of 7.03.
“Genetics has this interesting force, especially in MIT biology. The department’s historical — and active — role in genetics research ties directly into the way the course is taught,” Lee says. “Biology is about logic, scientific reasoning, and posing the right questions.”
A few years later, as a teaching assistant for class 7.002 (Fundamentals of Experimental Molecular Biology), he came to value how much care MIT biology professors take in presenting the material for all offered courses.
“I really appreciate how much effort MIT professors put into their teaching,” Lee says. “As a TA, you realize the beauty of how the professors organize these things — because they’re teaching you in a specific way, and you can grasp the beauty of it — there’s a beauty in studying and finding the patterns in nature.”
An undertaking to apply
To attend MIT at all hadn’t exactly been a lifelong dream. In fact, it didn’t occur to Lee that he could or should apply until he represented South Korea at the 49th International Chemistry Olympiad, where he won a Gold Medal in 2017. There, he had the chance to speak with MIT alumni, as well as current and aspiring students. More than half of those aspiring students eventually enrolled, Lee among them.
“Before that, MIT was this nearly mythical institution, so that experience really changed my life,” Lee recalls. “I heard so many different stories from people with so many different backgrounds — all converging towards the same enthusiasm towards science.”
At the time, Lee was already attending medical school — a six-year undergraduate program in Korea — that would lead to a stable career in medicine. Attending MIT would involve both changing his career plans and uprooting his life, leaving all his friends and family behind.
His parents weren’t especially enthusiastic about his desire to study at MIT, so it was up to Lee to meet the application requirements. He woke up at 3 a.m. to find his own way to the only SAT testing site in South Korea — an undertaking he now recalls with a laugh. In just three months, he had gathered everything he needed; MIT was the only institution in the United States Lee applied to.
He arrived in Cambridge, Massachusetts, in 2018 but attended MIT only for a semester before returning to Korea for his two years of mandatory military service.
“During military service, my goal was to read as many papers as possible, because I wondered what topic of science I’m drawn to — and many of the papers I was reading were authored by people I recognized, people who taught biology at MIT,” Lee says. “I became really interested in cancer biology.”
Return to MIT
When he returned to campus, Lee pledged to do everything he could to meet with faculty and discuss their work. To that end, he joined the MIT Undergraduate Research Journal, allowing him to interview professors. He notes that most MIT faculty are enthusiastic about being contacted by undergraduate students.
Stateside, Lee also reached out to Michael Lawrence, an assistant professor of pathology at Harvard Medical School and assistant geneticist at Mass General Cancer Center, about a preprint concerning APOBEC, an enzyme Lee had studied at Seoul National University. Lawrence’s lab was looking into APOBEC and cancer evolution — and the idea that the enzyme might drive drug resistance to cancer treatment.
“Since he joined my lab, I’ve been absolutely amazed by his scientific talents,” Lawrence says. “Hanjun’s scientific maturity and achievements are extremely rare, especially in an undergraduate student.”
Lee has made new discoveries from genomic data and was involved in publishing a paper in Molecular Cell and a paper in Nature Genetics. In the latter, the lab identified the source of background noise in chromosome conformation capture experiments, a technique for analyzing chromatin in cells.
Lawrence thinks Lee “is destined for great leadership in science.” In the meantime, Lee has gained valuable insights into how much work these types of achievements require.
“Doing research has been rewarding, but it also taught me to appreciate that science is almost 100 percent about failures,” Lee says. “It is those failures that end up leading you to the path of success.”
Widening the scope
Lee’s personal motto is that to excel in a specific field, one must have a broad sense of what the entire field looks like, and suggests other budding scientists enroll in courses distant from their research area. He also says it was key to see his peers as collaborators rather than competitors, and that each student will excel in their own unique way.
“Your MIT experience is defined by interactions with others,” Lee says. “They will help identify and shape your path.”
For his accomplishments, Lee was recently named an American Association for Cancer Research Undergraduate Scholar. Last year, he also spoke at the Gordon Research Conference on Cell Growth and Proliferation about his work on the retinoblastoma gene product RB.
Encouraged by positive course evaluations during his time as a TA, Lee hopes to inspire other students in the future through teaching. Lee has recently decided to pursue a PhD in cancer biology at Harvard Medical School, although his interests remain broad.
“I want to explore other fields of biology as well,” he says. “I have so many questions that I want to answer.”
Although initially resistant, Lee’s mother and father are now “immensely proud to be MIT parents” and will be coming to Cambridge in May to celebrate Lee’s graduation.
“Throughout my years here, they’ve been able to see how I’ve changed,” he says. “I don’t think I’m a great scientist, yet, but I now have some sense of how to become one.”

For science — and the scientists who practice it — to succeed, it must be shared. That’s why members of the MIT community recently gathered to learn about the research of eight postdocs from across the country for the second annual Catalyst Symposium, an event co-sponsored by the Department of Biology and The Picower Institute for Learning and Memory.
The eight Catalyst Fellows came to campus as part of an effort to increase engagement between MIT scholars and postdocs excelling in their respective fields from traditionally underrepresented backgrounds in science. The three-day symposium included panel discussions with faculty and postdocs, one-on-one meetings, social events, and research talks from the Catalyst Fellows.
“I love the name of this symposium because we’re all, of course, eager to catalyze advancements in our professional lives, in science, and to move forward faster by lowering activation barriers,” says MIT Biology Department Head Amy Keating. “I feel we can’t afford to do science with only part of the talent pool, and I don’t think people can do their best work when they are worried about whether they belong.”
The cohort of 2024 Catalyst Fellows included: Chloé Baron from Boston Children’s Hospital; Maria Cecília Canesso from The Rockefeller University; Kiara Eldred from the University of Washington School of Medicine; Caitlin Kowalski from the University of Oregon; Fabián Morales-Polanco from Stanford University; Kali Pruss from the Washington University School of Medicine in St. Louis; Rodrigo Romero from Memorial Sloan Kettering Cancer Center; and Zuri Sullivan from Harvard University.
Romero, who received his PhD from MIT working in the Jacks Lab at the Koch Institute, said that it was “incredible to see so many familiar faces,” but he spent the Symposium lunch chatting with new students in his old lab.
“Especially having been trained to think differently after MIT, I can now reach out to people that I didn’t as a graduate student, and make connections that I didn’t think about before,” Romero says.
He presented his work on lineage plasticity in the tumor microenvironment. Lineage plasticity is a hallmark of tumor progression but also occurs during normal development, such as wound healing.
As for the general mission of the symposium, Romero agreed with Keating.
“Trying to lower the boundary for other people to actually have a chance to do academic research in the future is important,” Romero says.
The Catalyst Symposium is aimed at early-career scientists who foresee a path in academia. Of the 2023 Catalyst Fellows, one has already secured a faculty position. Starting in September 2024, Shan Maltzer will be an assistant professor at Vanderbilt University in the Department of Pharmacology and the Vanderbilt Brain Institute studying mechanisms of somatosensory circuit assembly, development, and function.
Another aim of the Catalyst Symposium is to facilitate collaborations and strengthen existing relationships. Sullivan, an immunologist and molecular neuroscientist who presented on the interactions between the immune system and the brain, is collaborating with Sebastian Lourido, an Associate Professor of Biology and Core Member of the Whitehead Institute. Lourido’s studies include pathogens such as Toxoplasma gondii, which is known to alter the behavior of infected rodents. In the long term, Sullivan hopes to bridge research in immunology and neuroscience — for instance by investigating how infection affects behavior. She has observed that two rodents experiencing illness will huddle together in a cage, whereas an unafflicted rodent and an ill one will generally avoid each other when sharing the same space.
Pruss presented research on the interactions between the gut microbiome and the environment, and how they may affect physiology and fetal development. Kowalski discussed the relationship between fungi residing on our bodies and human health. Beyond the opportunity to deliver talks, both agreed that the small group settings of the three-day event were rewarding.
“The opportunity to meet with faculty throughout the symposium has been invaluable, both for finding familiar faces and for establishing friendly relationships,” Pruss says. “You don’t have to try to catch them when you’re running past them in the hallway.”
Eldred, who studies cell fate in the human retina, says she was excited about the faculty panels because they allowed her to ask faculty about fundamental aspects of recruiting for their labs, like bringing in graduate students.
Kowalski also says she enjoyed interfacing with so many new ideas — the spread of scientific topics from among the cohort of speakers extended beyond those she usually interacts with.
Mike Laub, Professor of Biology and HHMI Investigator, and Yadira Soto-Feliciano, Assistant Professor of Biology and Intramural Faculty at the Koch Institute, were on the symposium’s planning committee, along with Diversity, Equity, and Inclusion Officer Hallie Dowling-Huppert. Laub hopes the symposium will continue to be offered annually; next year’s Catalyst Symposium is already scheduled to take place in early May.
“I thought this year’s Catalyst Symposium was another great success. The talks from the visiting Fellows featured some amazing science from a wide range of fields,” Laub says. “I also think it’s fair to say that their interactions with the faculty, postdocs, and students here generated a lot of excitement and energy in our community, which is exactly what we hoped to accomplish with this symposium.”

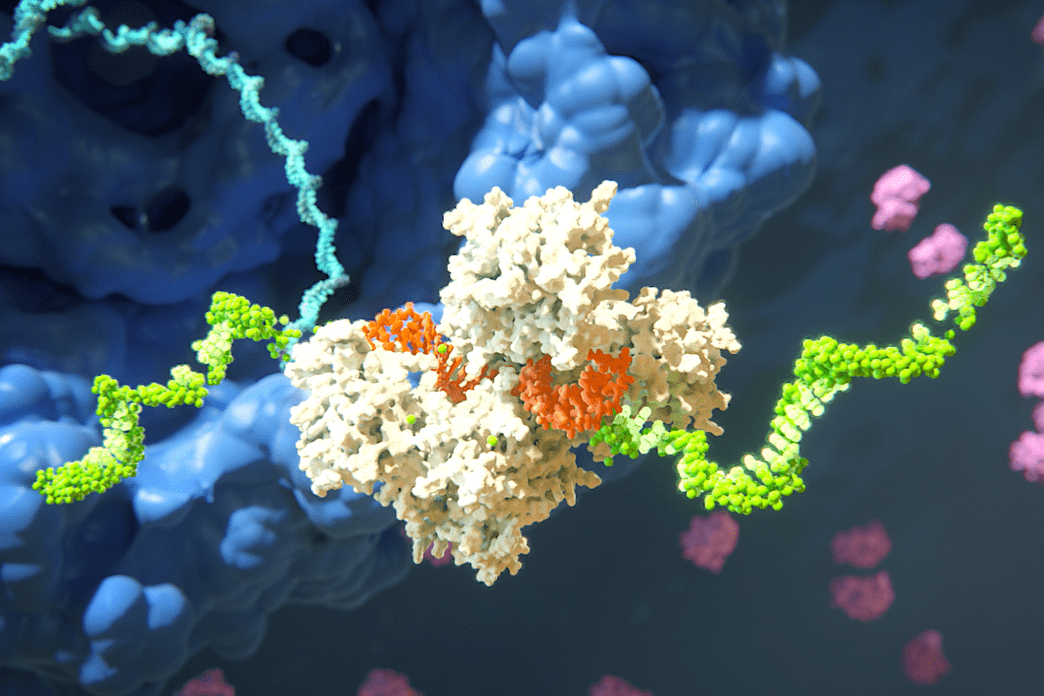
There are many hurdles to clear before a research discovery becomes a life-changing treatment for patients. That’s especially true when the treatments being developed represent an entirely new class of medicines. But overcoming those obstacles can revolutionize our ability to treat diseases.
Few companies exemplify that process better than Alnylam Pharmaceuticals. Alnylam was founded by a group of MIT-affiliated researchers who believed in the promise of a technology — RNA interference, or RNAi.
The researchers had done foundational work to understand how RNAi, which is a naturally occurring process, works to silence genes through the degradation of messenger RNA. But it was their decision to found Alnylam in 2002 that attracted the funding and expertise necessary to turn their discoveries into a new class of medicines. Since that decision, Alnylam has made remarkable progress taking RNAi from an interesting scientific discovery to an impactful new treatment pathway.
Today Alnylam has five medicines approved by the U.S. Food and Drug Administration (one Alnylam-discovered RNAi therapeutic is licensed to Novartis) and a rapidly expanding clinical pipeline. The company’s approved medicines are for debilitating, sometimes fatal conditions that many patients have grappled with for decades with few other options.
The company estimates its treatments helped more than 5,000 patients in 2023 alone. Behind that number are patient stories that illustrate how Alnylam has changed lives. A mother of three says Alnylam’s treatments helped her take back control of her life after being bed-ridden with attacks associated with the rare genetic disease acute intermittent porphyria (AIP). Another patient reported that one of the company’s treatments helped her attend her daughter’s wedding. A third patient, who had left college due to frequent AIP attacks, was able to return to school.
These days Alnylam is not the only company developing RNAi-based medicines. But it is still a pioneer in the field, and the company’s founders — MIT Institute Professor Phil Sharp, Professor David Bartel, Professor Emeritus Paul Schimmel, and former MIT postdocs Thomas Tuschl and Phillip Zamore — see Alnylam as a champion for the field more broadly.
“Alnylam has published more than 250 scientific papers over 20 years,” says Sharp, who currently serves as chair of Alnylam’s scientific advisory board. “Not only did we do the science, not only did we translate it to benefit patients, but we also described every step. We established this as a modality to treat patients, and I’m very proud of that record.”
Pioneering RNAi development
MIT’s involvement in RNAi dates back to its discovery. Before Andrew Fire PhD ’83 shared a Nobel Prize for the discovery of RNAi in 1998, he worked on understanding how DNA was transcribed into RNA, as a graduate student in Sharp’s lab.
After leaving MIT, Fire and collaborators showed that double-stranded RNA could be used to silence specific genes in worms. But the biochemical mechanisms that allowed double-stranded RNA to work were unknown until MIT professors Sharp, Bartel, and Ruth Lehmann, along with Zamore and Tuschl, published foundational papers explaining the process. The researchers developed a system for studying RNAi and showed how RNAi can be controlled using different genetic sequences. Soon after Tuschl left MIT, he showed that a similar process could also be used to silence specific genes in human cells, opening up a new frontier in studying genes and ultimately treating diseases.
“Tom showed you could synthesize these small RNAs, transfect them into cells, and get a very specific knockdown of the gene that corresponded to that the small RNAs,” Bartel explains. “That discovery transformed biological research. The ability to specifically knockdown a mammalian gene was huge. You could suddenly study the function of any gene you were interested in by knocking it down and seeing what happens. … The research community immediately started using that approach to study the function of their favorite genes in mammalian cells.”
Beyond illuminating gene function, another application came to mind.
“Because almost all diseases are related to genes, could we take these small RNAs and silence genes to treat patients?” Sharp remembers wondering.
To answer the question, the researchers founded Alnylam in 2002. (They recruited Schimmel, a biotech veteran, around the same time.) But there was a lot of work to be done before the technology could be tried in patients. The main challenge was getting RNAi into the cytoplasm of the patients’ cells.
“Through work in Dave Bartel and Phil Sharp’s lab, among others, it became evident that to make RNAi into therapies, there were three problems to solve: delivery, delivery, and delivery,” says Alnylam Chief Scientific Officer Kevin Fitzgerald, who has been with the company since 2005.
Early on, Alnylam collaborated with MIT drug delivery expert and Institute Professor Bob Langer. Eventually, Alnylam developed the first lipid nanoparticles (LNPs) that could be used to encase RNA and deliver it into patient cells. LNPs were later used in the mRNA vaccines for Covid-19.
“Alnylam has invested over 20 years and more than $4 billion in RNAi to develop these new therapeutics,” Sharp says. “That is the means by which innovations can be translated to the benefit of society.”
From scientific breakthrough to patient bedside
Alnylam received its first FDA approval in 2018 for treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis, a rare and fatal disease. It doubled as the first RNAi therapeutic to reach the market and the first drug approved to treat that condition in the United States.
“What I keep in mind is, at the end of the day for certain patients, two months is everything,” Fitzgerald says. “The diseases that we’re trying to treat progress month by month, day by day, and patients can get to a point where nothing is helping them. If you can move their disease by a stage, that’s huge.”
Since that first treatment, Alnylam has updated its RNAi delivery system — including by conjugating small interfering RNAs to molecules that help them gain entry to cells — and earned approvals to treat other rare genetic diseases along with high cholesterol (the treatment licensed to Novartis). All of those treatments primarily work by silencing genes that encode for the production of proteins in the liver, which has proven to be the easiest place to deliver RNAi molecules. But Alnylam’s team is confident they can deliver RNAi to other areas of the body, which would unlock a new world of treatment possibilities. The company has reported promising early results in the central nervous system and says a phase one study last year was the first RNAi therapeutic to demonstrate gene silencing in the human brain.
“There’s a lot of work being done at Alnylam and other companies to deliver these RNAis to other tissues: muscles, immune cells, lung cells, etc.,” Sharp says. “But to me the most interesting application is delivery to the brain. We think we have a therapeutic modality that can very specifically control the activity of certain genes in the nervous system. I think that’s extraordinarily important, for diseases from Alzheimer’s to schizophrenia and depression.”
The central nervous system work is particularly significant for Fitzgerald, who watched his father struggle with Parkinson’s.
“Our goal is to be in every organ in the human body, and then combinations of organs, and then combinations of targets within individual organs, and then combinations of targets within multi-organs,” Fitzgerald says. “We’re really at the very beginning of what this technology is going do for human health.”
It’s an exciting time for the RNAi scientific community, including many who continue to study it at MIT. Still, Alnylam will need to continue executing in its drug development efforts to deliver on that promise and help an expanding pool of patients.
“I think this is a real frontier,” Sharp says. “There’s major therapeutic need, and I think this technology could have a huge impact. But we have to prove it. That’s why Alnylam exists: to pursue new science that unlocks new possibilities and discover if they can be made to work. That, of course, also why MIT is here: to improve lives.”


When entering the office of Building 68’s manager, you will likely be greeted first with an amiable nose boop and wagging tail from Shadow, a four-year-old black lab, followed by a warm welcome from the office’s other occupant, John Fucillo. Fucillo is an animal lover, and Shadow is the gentlest of roughly nine dogs and one Siamese cat he’s taken care of throughout his life. Fortunately for MIT Biology, Shadow is not the only lab Fucillo cares for.
A Boston area local, Fucillo spent two years working at Revere Beach, then learned skills as an auto mechanic, and later completed an apprenticeship with the International Brotherhood of Electrical Workers. In 1989, Fucillo came to MIT Biology and says he couldn’t be happier.
As Building 68’s manager, Environment, Health & Safety coordinator, and Chemical Hygiene Officer, Fucillo’s goal is to make workflows “easier, less expensive, more desirable, and more comfortable.”
Fucillo was key for the Department’s successful move into its new home when Building 68 was completed in 1994, according to Mitchell Galanek, MIT Radiation Protection Officer and Fucillo’s colleague for over 30 years.
Throughout his time as a building manager, Fucillo has decreased routine spending and increased sustainability. He lowered the cost of lab coats by a whopping 92%–from $2,600 to $200–with just one phone call to North Star, the building’s uniform/linens provider. Auditing the building’s plastic waste generation inspired the institute-wide MIT Lab Plastics Recycling Program, which now serves over 200 labs across campus. More than 50,000 lbs of plastic have been recycled in the last four years alone.
“John is not a cog in the wheel, but an integral part of the whole system,” says Anthony Fuccione, Technical Instructor and Manager of the Biology Teaching lab.
Fucillo says one of his favorite parts of the job is chatting with researchers and helping them achieve their goals. He reportedly clocks about 10,000 steps per day on campus, responding to requests from labs, collaborating with colleagues, and connecting Biology to the institute’s Environment, Health, & Safety office.
“John is called upon — literally and figuratively — morning, noon, and night,” says Whitehead Professor of Molecular Genetics Monty Krieger. “He has had to become an expert in so very many areas to support staff, faculty, and students. His enormous success is due in part to his technical talents, in part to his genuine care for the welfare of his colleagues, and in part to his very special and caring personality.”
When MIT needed to comply with the EPA’s decree to improve safety standards across campus, Fucillo sat on the committees tasked with meeting those standards while avoiding undue burden on researchers, establishing the Environmental Health and Safety Management system in 2002.
“From a safety perspective, that was one of the most challenging things MIT had to go through–but it came out at the end a better, safer, place,” says John Collins, EHS Project Technician and friend and colleague to Fucillo for over 20 years.
Fucillo later co-led the initiative for a 2011 overhaul of MIT’s management of regulated medical waste (RMW), such as Petri dishes, blood, and needles. Fucillo volunteered to pilot a new approach in Building 68 — despite a lukewarm response to the proposal from other Biology EHS representatives, according to Galanek. This abundantly successful management system is now used by all MIT departments that generate RMW. It’s not only less expensive, but also does a better job at decontaminating waste than the previous management system.
“Anyone who has worked with John during his MIT career understands it is truly a privilege to partner with him,” Galanek says. “Not only does the work get done and done well, but you also gain a friend along the way.”
After consolidating a disparate group of individual lab assistants, Fucillo took on a supervisory role for the centralized staff tasked with cleaning glassware, preparing media, and ensuring consistency and sterility across Building 68 labs.
According to maintenance mechanic James “Jimmy” Carr, “you can’t find a better boss.”
“He’s just an easy-going guy,” says Karen O’Leary, who has worked with Fucillo for over 30 years. “My voice matters–I feel heard and respected by him.”
Although there are still many updates Fucillo hopes to see in Building 68, which will soon celebrate its 30th birthday, he is taking steps to cut back on his workload.
He recently began passing on his knowledge to Facilities Manager and EHS Coordinator Cesar Duarte, who joined the department in 2023.
“It’s been a pleasure working alongside John and learning about the substantial role and responsibility he’s had in the Biology department for the last three decades,” Duarte says. “Not only is John’s knowledge of Building 68 and the department’s history unparalleled, but his dedication to MIT and continued care and commitment to the health and well-being of the Biology community throughout his career are truly remarkable.”
As he winds down his time at MIT, Fucillo hopes to spend more time on music, one of his earliest passions, which began when he picked up an accordion in first grade. He still plays guitar and bass nearly every day. When he rocks out at home more often, he’ll be leaving behind the foundations of innovation, leadership, and respect in Building 68.