Place cells are well known to encode individual locations, but new experiments and analysis indicate that stitching together a “cognitive map” of a whole environment requires a broader ensemble of cells, aided by sleep, to build a richer network over several days, according to new research from the Wilson Lab.
David Orenstein | The Picower Institute for Learning and Memory
December 10, 2024
On the first day of your vacation in a new city your explorations expose you to innumerable individual places. While the memories of these spots (like a beautiful garden on a quiet side street) feel immediately indelible, it might be days before you have enough intuition about the neighborhood to direct a newer tourist to that same site and then maybe to the café you discovered nearby. A new study in mice by MIT neuroscientists at The Picower Insitute for Learning and Memory provides new evidence for how the brain forms cohesive cognitive maps of whole spaces and highlights the critical importance of sleep for the process.
Scientists have known for decades that the brain devotes neurons in a region called the hippocampus to remembering specific locations. So-called “place cells” reliably activate when an animal is at the location the neuron is tuned to remember. But more useful than having markers of specific spaces is having a mental model of how they all relate in a continuous overall geography. Though such “cognitive maps” were formally theorized in 1948, neuroscientists have remained unsure of how the brain constructs them. The new study in the December edition of Cell Reports finds that the capability may depend upon subtle but meaningful changes over days in the activity of cells that are only weakly attuned to individual locations, but that increase the robustness and refinement of the hippocampus’s encoding of the whole space. With sleep, the study’s analyses indicate, these “weakly spatial” cells increasingly enrich neural network activity in the hippocampus to link together these places into a cognitive map.
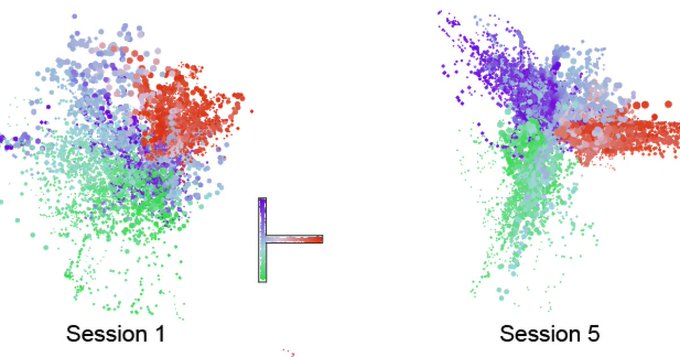
“On day 1, the brain doesn’t represent the space very well,” said lead author Wei Guo, a research scientist in the lab of senior author Matthew Wilson, Sherman Fairchild Professor in The Picower Institute and MIT’s Departments of Biology and Brain and Cognitive Sciences. “Neurons represent individual locations, but together they don’t form a map. But on day 5 they form a map. If you want a map, you need all these neurons to work together in a coordinated ensemble.”
Mice mapping mazes
To conduct the study, Guo and Wilson along with labmates Jie “Jack” Zhang and Jonathan Newman introduced mice to simple mazes of varying shapes and let them explore them freely for about half an hour a day for several days. Importantly, the mice were not directed to learn anything specific through the offer of any rewards. They just wandered. Previous studies have shown that mice naturally demonstrate “latent learning” of spaces from this kind of unrewarded experience after several days.
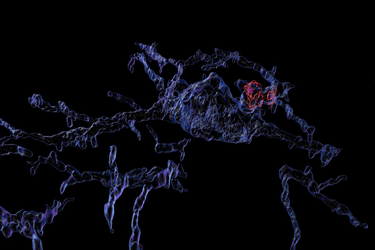
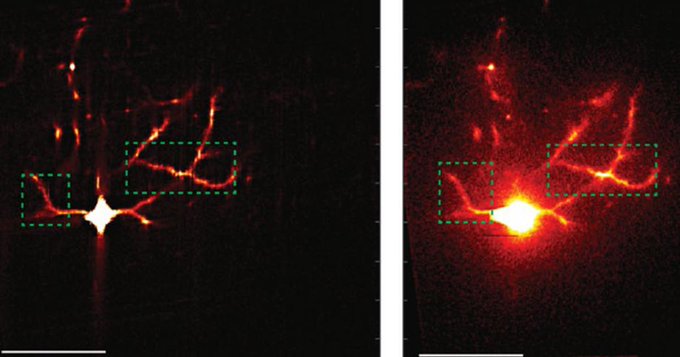
To understand how latent learning takes hold, Guo and his colleagues visually monitored hundreds of neurons in the CA1 area of the hippocampus by engineering cells to flash when a buildup of calcium ions made them electrically active. They not only recorded the neurons’ flashes when the mice were actively exploring, but also while they were sleeping. Wilson’s lab has shown that animals “replay” their previous journeys during sleep, essentially refining their memories by dreaming about their experiences.
Analysis of the recordings showed that the activity of the place cells developed immediately and remained strong and unchanged over several days of exploration. But this activity alone wouldn’t explain how latent learning or a cognitive map evolves over several days. So unlike in many other studies where scientists focus solely on the strong and clear activity of place cells, Guo extended his analysis to the more subtle and mysterious activity of cells that were not so strongly spatially tuned. Using an emerging technique called “manifold learning” he was able to discern that many of the “weakly spatial” cells gradually correlated their activity not with locations, but with activity patterns among other neurons in the network. As this was happening, Guo’s analyses showed, the network encoded a cognitive map of the maze that increasingly resembled the literal, physical space.
“Although not responding to specific locations like strongly spatial cells, weakly spatial cells specialize in responding to ‘‘mental locations,’’ i.e., specific ensemble firing patterns of other cells,” the study authors wrote. “If a weakly spatial cell’s mental field encompasses two subsets of strongly spatial cells that encode distinct locations, this weakly spatial cell can serve as a bridge between these locations.”
In other words, the activity of the weakly spatial cells likely stitches together the individual locations represented by the place cells into a mental map.
The need for sleep
Studies by Wilson’s lab and many others have shown that memories are consolidated, refined and processed by neural activity, such as replay, that occurs during sleep and rest. Guo and Wilson’s team therefore sought to test whether sleep was necessary for the contribution of weakly spatial cells to latent learning of cognitive maps.
To do this they let some mice explore a new maze twice during the same day with a three-hour siesta in between. Some of the mice were allowed to sleep but some were not. The ones that did showed a significant refinement of their mental map, but the ones that weren’t allowed to sleep showed no such improvement. Not only did the network encoding of the map improve, but also measures of the tuning of individual cells during showed that sleep helped cells become better attuned both to places and to patterns of network activity, so called “mental places” or “fields.”
Mental map meaning
The “cognitive maps” the mice encoded over several days were not literal, precise maps of the mazes, Guo notes. Instead they were more like schematics. Their value is that they provide the brain with a topology that can be explored mentally, without having to be in the physical space. For instance, once you’ve formed your cognitive map of the neighborhood around your hotel, you can plan the next morning’s excursion (e.g. you could imagine grabbing a croissant at the bakery you observed a few blocks west and then picture eating it on one of those benches you noticed in the park along the river).
Indeed, Wilson hypothesized that the weakly spatial cells’ activity may be overlaying salient non-spatial information that brings additional meaning to the maps (i.e. the idea of a bakery is not spatial, even if it’s closely linked to a specific location). The study, however, included no landmarks within the mazes and did not test any specific behaviors among the mice. But now that the study has identified that weakly spatial cells contribute meaningfully to mapping, Wilson said future studies can investigate what kind of information they may be incorporating into the animals’ sense of their environments. We seem to intuitively regard the spaces we inhabit as more than just sets of discrete locations.
“In this study we focused on animals behaving naturally and demonstrated that during freely exploratory behavior and subsequent sleep, in the absence of reinforcement, substantial neural plastic changes at the ensemble level still occur,” the authors concluded. “This form of implicit and unsupervised learning constitutes a crucial facet of human learning and intelligence, warranting further in-depth investigations.”
The Freedom Together Foundation, The Picower Institute for Learning and Memory and the National Institutes of Health funded the study.