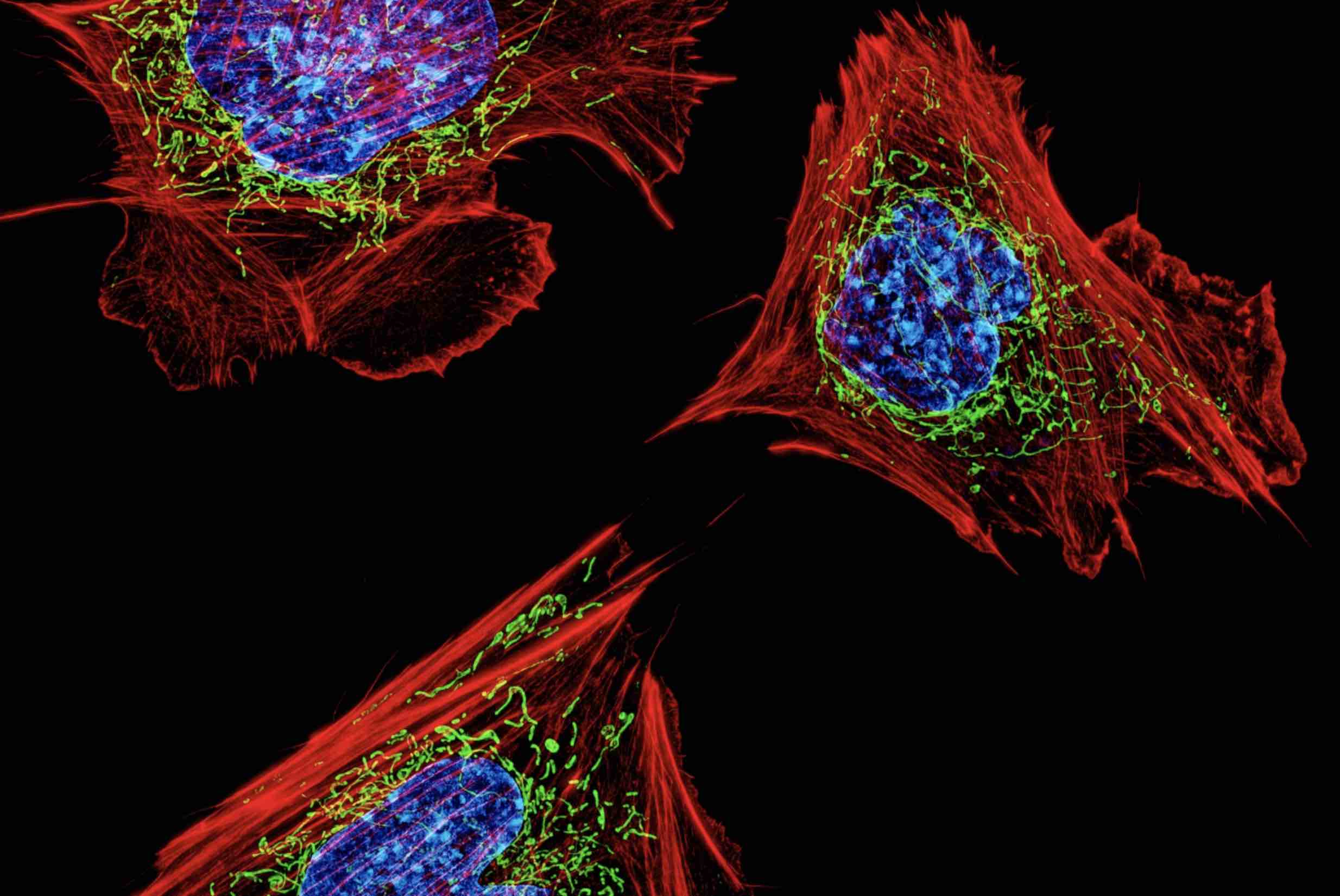
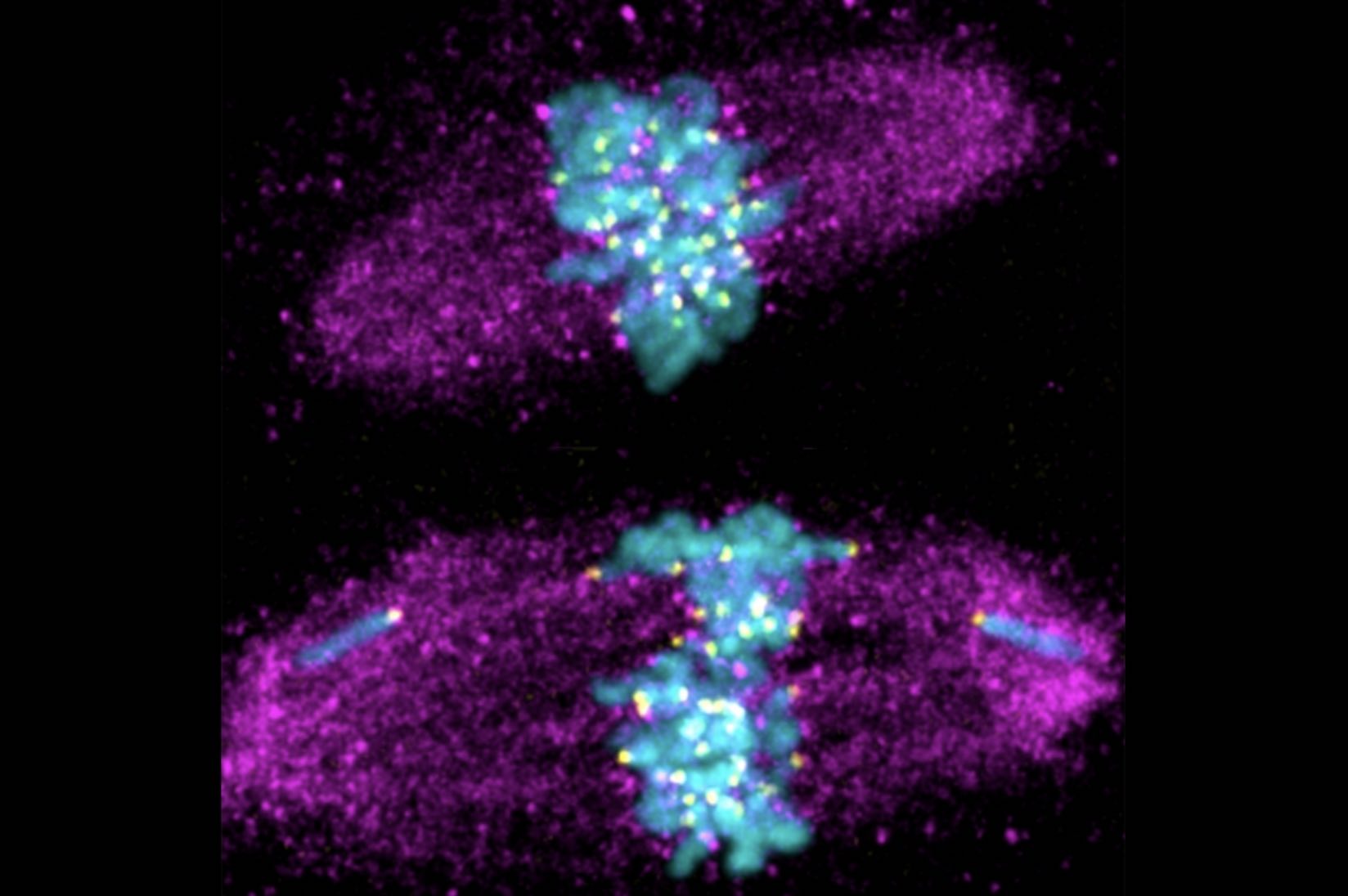
The clinically-trained cell biologist exploits the liver’s unique capacities in search of new medical applications.
Grace van Deelen | Department of Biology
December 15, 2021
Why is the liver the only human organ that can regenerate? How does it know when it’s been injured? What can our understanding of the liver contribute to regenerative medicine? These are just some of the questions that new assistant professor of biology Kristin Knouse and her lab members are asking in their research at the Koch Institute for Integrative Cancer Research. Knouse sat down to discuss why the liver is so unique, what lessons we might learn from the organ, and what its regeneration might teach us about cancer.
Q: Your lab is interested in questions about how body tissues sense and respond to damage. What is it about the liver that makes it a good tool to model those questions?
A: I’ve always felt that we, as scientists, have so much to gain from treasuring nature’s exceptions, because those exceptions can shine light onto a completely unknown area of biology and provide building blocks to confer such novelty to other systems. When it comes to organ regeneration in mammals, the liver is that exception. It is the only solid organ that can completely regenerate itself. You can damage or remove over 75 percent of the liver and the organ will completely regenerate in a matter of weeks. The liver therefore contains the instructions for how to regenerate a solid organ; however, we have yet to access and interpret those instructions. If we could fully understand how the liver is able to regenerate itself, perhaps one day we could coax other solid organs to do the same.
There are some things we already know about liver regeneration, such as when it begins, what genes are expressed, and how long it takes. However, we still don’t understand why the liver can regenerate but other organs cannot. Why is it that these fully differentiated liver cells — cells that have already assumed specialized roles in the liver — can re-enter the cell cycle and regenerate the organ? We don’t have a molecular explanation for this. Our lab is working to answer this fundamental question of cell and organ biology and apply our discoveries to unlock new approaches for regenerative medicine. In this regard, I don’t necessarily consider myself exclusively a liver biologist, but rather someone who is leveraging the liver to address this much broader biological problem.
Q: As an MD/PhD student, you conducted your graduate research in the lab of the late Professor Angelika Amon here at MIT. How did your work in her lab lead to an interest in studying the liver’s regenerative capacities?
A: What was incredible about being in Angelika’s lab was that she had an interest in almost everything and gave me tremendous independence in what I pursued. I began my graduate research in her lab with an interest in cell division, and I was doing experiments to observe how cells from different mammalian tissues divide. I was isolating cells from different mouse tissues and then studying them in culture. In doing that, I found that when the cells were isolated and grown in a dish they could not segregate their chromosomes properly, suggesting that the tissue environment was essential for accurate cell division. In order to further study and compare these two different contexts — cells in a tissue versus cells in culture — I was keen to study a tissue in which I could observe a lot of cells undergoing cell division at the same time.
So I thought back to my time in medical school, and I remembered that the liver has the ability to completely regenerate itself. With a single surgery to remove part of the liver, I could stimulate millions of cells to divide. I therefore began exploiting liver regeneration as a means of studying chromosome segregation in tissue. But as I continued to perform surgeries on mice and watch the liver rapidly regenerate itself, I couldn’t help but become absolutely fascinated by this exceptional biological process. It was that fascination with this incredibly unique but poorly understood phenomenon — alongside the realization that there was a huge, unmet medical need in the area of regeneration — that convinced me to dedicate my career to studying this.
Q: What kinds of clinical applications might a better understanding of organ regeneration lead to, and what role do you see your lab playing in that research?
A: The most proximal medical application for our work is to confer regenerative capacity to organs that are currently non-regenerative. As we begin to achieve a molecular understanding of how and why the liver can regenerate, we put ourselves in a powerful position to identify and surmount the barriers to regeneration in non-regenerative tissues, such as the heart and nervous system. By answering these complementary questions, we bring ourselves closer to the possibility that, one day, if someone has a heart attack or a spinal cord injury, we could deliver a therapy that stimulates the tissue to regenerate itself. I realize that may sound like a moonshot now, but I don’t think any problem is insurmountable so long as it can be broken down into a series of tractable questions.
Beyond regenerative medicine, I believe our work studying liver regeneration also has implications for cancer. At first glance this may seem counterintuitive, as rapid regrowth is the exact opposite of what we want cancer cells to do. However, the reality is that the majority of cancer-related deaths are attributable not to the rapidly proliferating cells that constitute primary tumors, but rather to the cells that disperse from the primary tumor and lie dormant for years before manifesting as metastatic disease and creating another tumor. These dormant cells evade most of the cancer therapies designed to target rapidly proliferating cells. If you think about it, these dormant cells are not unlike the liver: they are quiet for months, maybe years, and then suddenly awaken. I hope that as we start to understand more about the liver, we might learn how to target these dormant cancer cells, prevent metastatic disease, and thereby offer lasting cancer cures.