
Graduate student Zhaoqi Li investigates how cancer cells grow by harnessing exceptional chemical reactions
Justin Chen
January 11, 2018
Cancer cells use extreme measures to fuel their growth. In fact, researchers like Zhaoqi Li, a third-year graduate student, witness chemical reactions in these cells that would be impossible in the context of normal cells. In a petri dish, normal cells stop dividing once they cover the bottom of the dish and fit neatly together like mosaic tiles. In contrast, cancer cells continue to proliferate and pile haphazardly into small mounds. Within the human body, this abnormal growth — when combined with the spread of cancer cells throughout the body — interferes with organ function and causes death.
Li, a member of Professor Matthew Vander Heiden’s lab located in the Koch Institute, studies cancer metabolism. His work describes the chemical reactions cancer cells use to create energy and materials to make new cells such as membranes, proteins, and DNA. By tracking the flow of nutrients through cancer cells, Li and his labmates are learning how such cells change their metabolism to stimulate growth. These insights will help scientists develop new ways to treat the disease.
Cell metabolism comprises all the chemical reactions occurring in the cell, but researchers are particularly interested in a few reactions that aren’t required by normal cells but are critical for cancer growth. Stopping these reactions with drugs would disrupt the metabolism of cancer cells and hinder tumor development.
“Even though many people may not think of metabolism as a treatment target for cancer, this strategy has been used unwittingly for a long time,” Li says. “Many chemotherapies, such as antifolates, were originally used by doctors without knowing exactly how they worked. Since then, we’ve discovered that those treatments target metabolic pathways. By understanding the details of cancer metabolism we are hoping to design drugs in a more rational way.”
– –
Li might never have joined the Vander Heiden lab or studied cancer metabolism were it not for the unique structure of graduate training at MIT.
During their first year at MIT, graduate students are required to take four classes. Unlike their counterparts at many other PhD programs, they do not work in laboratories until their second semester. This allows students to focus initially on coursework — covering biochemistry, genetics, and research methodology — designed to build a foundation of knowledge. As a result, students discover new interests and develop the confidence to move out of their comfort zones. When it comes time to select a lab, they can choose from 56 spread across six locations, spanning a wide breadth of biological research.
Li could study how the brain forms memories, interpret X-rays to deduce protein structure, or even build miniature organs for drug testing. Before making his decision, he rotated in three laboratories. During each month-long rotation, he performed a small project allowing him to experience the culture of the lab and learn more about its research.
“The first two labs I visited were studying topics I was familiar with and thought were interesting,” he says. “But when I visited the Vander Heiden lab it was so different and caught me off guard. That’s why I eventually joined, even though I had never imagined myself working in a metabolism lab before.”
– –
Although he is new to the community of researchers specializing in metabolism, Li has long known that he wanted to interact with the world through science. As an immigrant who moved from China to southern Tennessee at the age of six, Li struggled to learn English and began to view science as a universal language that transcended culture.
“My parents were also non-native speakers and the English as a Second Language classes in my elementary school were geared towards Spanish speakers, so I had a really hard time,” Li says. “I joke that the only reason I passed the first grade was because I was good at math.”
Li’s contrasting relationship with science and English continued as an undergraduate at Columbia University. There he majored in biochemistry and also studied literature of the Western Canon to fulfill his general degree requirements.
“I took four semesters worth of classes that started with Plato and ended with Virginia Woolf,” he says, “It was an eye-opening experience, but I never really loved it. I found biology more intuitive because it doesn’t rely on being familiar with a specific cultural lens. Most every society in the world values the scientific method to some extent.”
Li began working in a lab during his sophomore year at Columbia. To his surprise, he was mentored by a professor who valued his input and encouraged creative thinking. Li’s supervisor also introduced him to basic science — a type of research driven not by the desire to find a specific answer or cure, but by curiosity and the need to better understand the natural world.
– –
During his second semester rotation at MIT, Li searched for similarly open-minded environments, and was attracted to cancer metabolism because the field was relatively young.
“In other more established areas of biology, if you have a question someone has probably answered it in some capacity,” Li says. “The Vander Heiden lab was using new techniques so there was a lot of space to explore. Many questions I asked — even during my initial rotation — didn’t have an answer, which was exciting.”
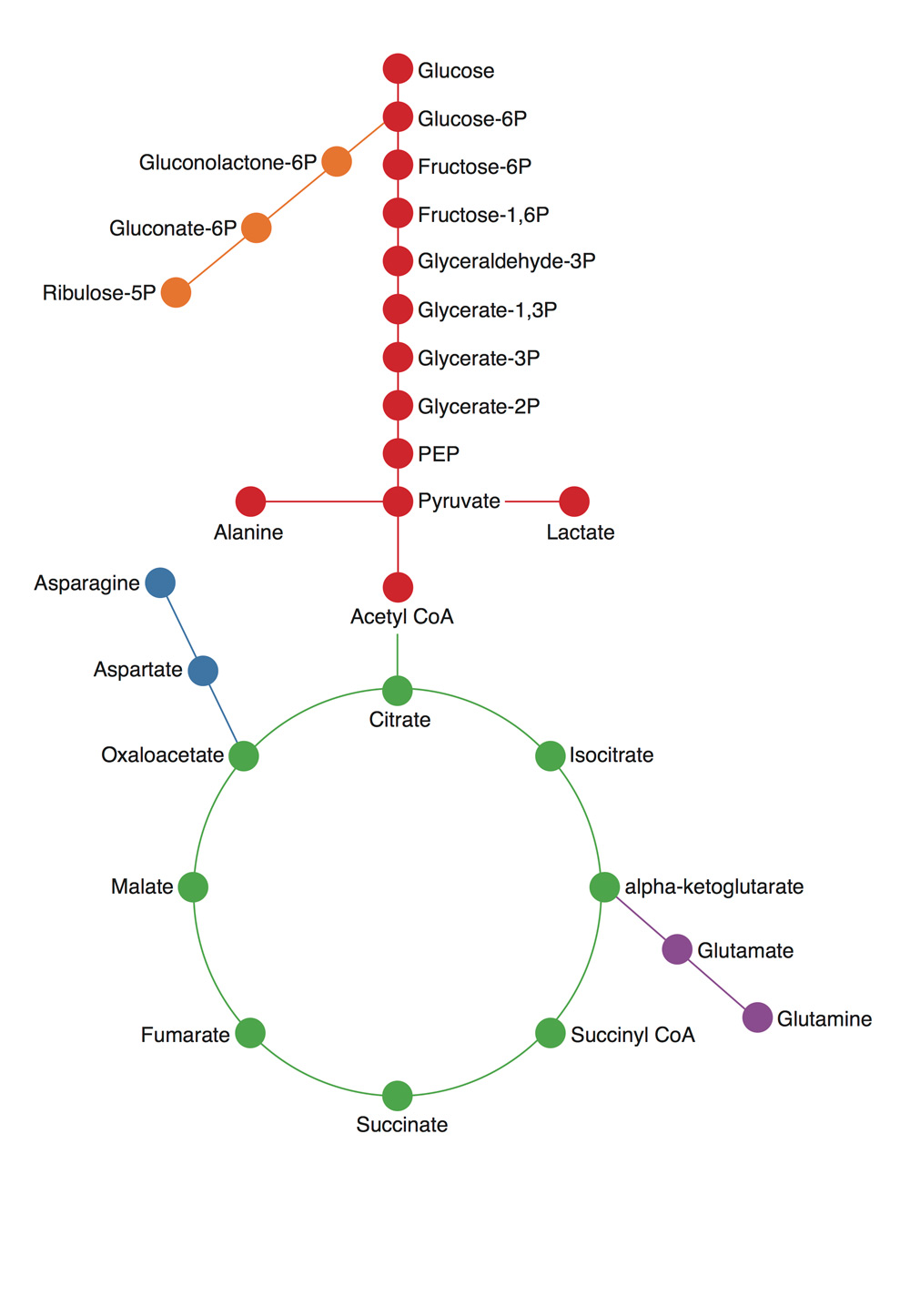
The great challenge confronting the metabolism field is translating decades’ worth of research on enzymes — proteins that manage chemical reactions — from the test tube to the cell and human body. By studying enzymes individually in the controlled setting of test tubes, researchers have documented almost all the chemical reactions that occur in the cell. When combined, these reactions look like a giant subway map where each stop, indicated by a dot, is a different molecule, and the line between stops represents a chemical reaction where atoms are added or subtracted. Some pathways are a straight line but others have nodes or intersections where a molecule can take part in several different reactions. Other pathways are circular where the molecule that starts the pathway is remade at the end so that the line circles back on itself.
Despite the ability to study chemical reactions in a test tube, scientists have struggled to understand what is actually happening in the complex environment of cells, which coordinate millions of reactions that not only affect each other, but are also influenced by outside stresses like nutrient deprivation.
To Li, using the metabolism map to figure out what chemical reactions are occurring and how atoms are moving through the cell is like using a subway map to track how people are traveling through a city.
“The map describes all the possible routes people could take,” Li says, “but you have to track the passengers to figure out where they are actually going. You could imagine people commuting into the city during the week and going to entirely different places on the weekend. There are a lot of different patterns of movement that you can’t infer just from looking at a map.”
To analyze what chemical reactions are occurring in the cell, Li utilizes cutting edge technology to track carbon atoms — an essential element that is required to build all components of the cell. By tagging carbon with an extra neutron, Li makes the experimentally altered atom heavier and distinguishable from naturally occurring carbon in the cell. Feeding cells nutrients like glucose made with heavy carbons allows Li to compare how molecules are broken down and used by normal and cancerous cells.
 “Returning to the subway map analogy, this labeling technique is similar to not only being inside the subway, but also giving everyone in Downtown Boston a red shirt,” Li says. “After 12 hours, we can look at the rest of the city. If we see a lot of red shirts in Allston, we would know that this particular route is really popular.”
“Returning to the subway map analogy, this labeling technique is similar to not only being inside the subway, but also giving everyone in Downtown Boston a red shirt,” Li says. “After 12 hours, we can look at the rest of the city. If we see a lot of red shirts in Allston, we would know that this particular route is really popular.”
In the case of glucose, Li and his labmates observed that normal cells break down the sugar to release energy and heavy carbons in the form of carbon dioxide. In contrast, cancer cells alter their metabolism so that the heavy carbons originally found in glucose are used to build new parts of the cells that are required for cancer cells to grow, such as membranes, DNA, and proteins.
Li’s observations demonstrate how cancer cells sustain abnormal growth by accumulating carbon. For his thesis project, Li has chosen to investigate one of the main tricks cancer cells use to hoard carbon atoms: a process known as carbon fixation. This type of chemical reaction, originally studied in plants performing photosynthesis, attaches carbon dioxide to other molecules. Li’s initial findings suggest that a protein, Malic Enzyme 1, helps cancer cells use carbon dioxide to build components required for growing and dividing.
“This is surprising,” he says, “because the textbook version of this enzyme actually catalyzes the reverse reaction in normal cells where carbon dioxide is removed from molecules. Malic Enzyme 1 is an example of how cancer performs remarkable chemical reactions — who would have thought that cancer cells use carbon like plants do?”
Li is at the beginning stages of his research, and can’t predict where his project will take him. His current goal is to determine how cancer cells react when they are missing Malic Enzyme 1. Such loss could slow growth, but Li will have to perform experiments to be sure, since cancer is a resourceful and elusive target.
Like a detour rerouting travelers around a closed metro stop, cancer cells may further contort their metabolism, taking advantage of little-used or still unidentified chemical reactions to maintain growth. In the face of such adaptability, Li and his labmates believe the best course of action is to be as curious as possible to understand as much as they can about how cancer works. Working together, they discuss confounding results, adjust hypotheses, and design new experiments.
“It’s really encouraging to be part of Matt’s lab and the Koch Institute in general where researchers take a basic science approach,” Li says. “We try to keep an open mind because there’s probably no single thing that cancer cells depend on. Everyone’s work builds together to form a cumulative understanding.”
