


Ferroptosis is an iron-dependent form of cell death with profound implications in human health and disease. In the context of cancer, the use of ferroptosis inducers to target subpopulations of highly metastatic and therapy-resistant cancer cells has garnered much excitement over the last few years. However, to gain a comprehensive understanding of the full therapeutic potential of ferroptosis, our research focuses on (i) uncovering the molecular factors affecting ferroptosis susceptibility, (ii) studying its impact on the tumor microenvironment, and (iii) developing innovative ways to modulate ferroptosis resistance in vivo. We employ a multidisciplinary approach, combining functional genomics, metabolomics, bioengineering, and a range of in vitro and in vivo models to advance our understanding in this domain and to translate our findings into effective therapies.

Sipping a beer on an early autumn evening, one might not consider that humans and yeast have been inextricably linked for thousands of years; winemaking, baking, and brewing all depend on budding yeast. Outside of baking and fermentation, researchers also use Saccharomyces cerevisiae, classified as a fungus, to study fundamental questions of cell biology.
Budding yeast gets its name from the way it multiplies. A daughter cell forms first as a swelling, protruding growth on the mother cell. The daughter cell projects further and further from the mother cell until it detaches as an independent yeast cell.
How do cells decide on a front and back? How do cells decode concentration gradients of chemical signals to orient in useful directions, or sense and navigate around physical obstacles? New Department of Biology faculty member Daniel “Danny” Lew uses the model yeast S. cerevisiae, and a non-model yeast with an unusual pattern of cell division, to explore these questions.
Q: Why is it useful to study yeast, and how do you approach the questions you hope to answer?
A: Humans and yeast are descended from a common ancestor, and some molecular mechanisms developed by that ancestor have been around for so long that yeast and mammals often use the same mechanisms. Many cells develop a front and migrate or grow in a particular direction, like the axons in our nervous system, using similar molecular mechanisms to those of yeast cells orienting growth towards the bud.
When I started my lab, I was working on cell cycle control, but I’ve always been interested in morphogenesis and the cell biology of how cells change shape and decide to do different things with different parts of themselves. Those mechanisms turn out to be conserved between yeast and humans.
But some things are very different about fungal and animal cells. One of the differences is the cell wall and what fungal cells have to do to deal with the fact that they have a cell wall.
Fungi are inflated by turgor pressure, which pushes their membranes against the rigid cell wall. This means they’ll die if there is any hole in the cell wall, which would be expected to happen often as cells remodel the wall in order to grow. We’re interested in understanding how fungi sense when any weak spots appear in the wall and repair them before those weak spots become dangerous.
Yeast cells, like most fungi, also mate by fusing with a partner. To succeed, they must do the most dangerous thing in the fungal life cycle: get rid of the cell wall at the point of contact to allow fusion. That means they must be precise about where and when they remove the wall. We’re fascinated to understand how they know it is safe to remove the wall there, and nowhere else.
We take an interdisciplinary approach. We’ve used genetics, biochemistry, cell biology, and computational biology to try and solve problems in the past. There’s a natural progression: observation and genetic approaches tend to be the first line of attack when you know nothing about how something works. As you learn more, you need biochemical approaches and, eventually, computational approaches to understand exactly what mechanism you’re looking at.
I’m also passionate about mentoring, and I love working with trainees and getting them fascinated by the same problems that fascinate me. I’m looking to work with curious trainees who love addressing fundamental problems.
Q: How does yeast decide to orient a certain way — toward a mating partner, for example?
A: We are still working on questions of how cells analyze the surrounding environment to pick a direction. Yeast cells have receptors that sense pheromones that a mating partner releases. What is amazing about that is that these cells are incredibly small, and pheromones are released by several potential partners in the neighborhood. That means yeast cells must interpret a very confusing landscape of pheromone concentrations. It’s not apparent how they manage to orient accurately toward a single partner.
That got me interested in related questions. Suppose the cell is oriented toward something that isn’t a mating partner. The cell seems to recognize that there’s an obstacle in the way, and it can change direction to go around that obstacle. This is how fungi get so good at growing into things that look very solid, like wood, and some fungi can even penetrate Kevlar vests.
If they recognize an obstacle, they have to change directions and go around it. If they recognize a mating partner, they have to stick with that direction and allow the cell wall to get degraded. How do they know they’ve hit an obstacle? How do they know a mating partner is different from an obstacle? These are the questions we’d like to understand.
Q: For the last couple of years, you’ve also been studying a budding yeast that forms multiple buds when it reproduces instead of just one. How did you come across it, and what questions are you hoping to explore?
A: I spent several years trying to figure out why most yeasts make one bud and only one bud, which I think is related to the question of why migrating cells make one and only one front. We had what we thought was a persuasive answer to that, so seeing a yeast completely disobey that and make as many buds as it felt like was a shock, which got me intrigued.
We started working on it because my colleague, Amy Gladfelter, had sampled the waters around Woods Hole, Massachusetts. When she saw this specimen under a microscope, she immediately called me and said, “You have to look at this.”
A question we’re very intrigued by is if the cell makes five, seven, or 12 buds simultaneously, how do they divide the mother cell’s material and growth capacity five, seven, or 12 ways? It looks like all of the buds grow at the same rate and reach about the same size. One of our short-term goals is to check whether all the buds really get to exactly the same size or whether they are born unequal.
And we’re interested in more than just growth rate. What about organelles? Do you give each bud the same number of mitochondria, nuclei, peroxisomes, and vacuoles? That question will inevitably lead to follow-up questions. If each bud has the same number of mitochondria, how does the cell measure mitochondrial inheritance to do that? If they don’t have the same amount, then buds are each born with a different complement and ratio of organelles. What happens to buds if they have very different numbers of organelles?
As far as we can tell, every bud gets at least one nucleus. How the cell ensures that each bud gets a nucleus is a question we’d also very much like to understand.
We have molecular candidates because we know a lot about how model yeasts deliver nuclei, organelles, and growth materials from the mother to the single bud. We can mutate candidate genes and see if similar molecular pathways are involved in the multi-budding yeast and, if so, how they are working.
It turns out that this unconventional yeast has yet to be studied from the point of view of basic cell biology. The other thing that intrigues me is that it’s a poly-extremophile. This yeast can survive under many rather harsh conditions: it’s been isolated in Antarctica, from jet engines, from all kinds of plants, and of course from the ocean as well. An advantage of working with something so ubiquitous is we already know it’s not toxic to us under almost any circumstances. We come into contact with it all the time. If we learn enough about its cell biology to begin to manipulate it, then there are many potential applications, from human health to agriculture.
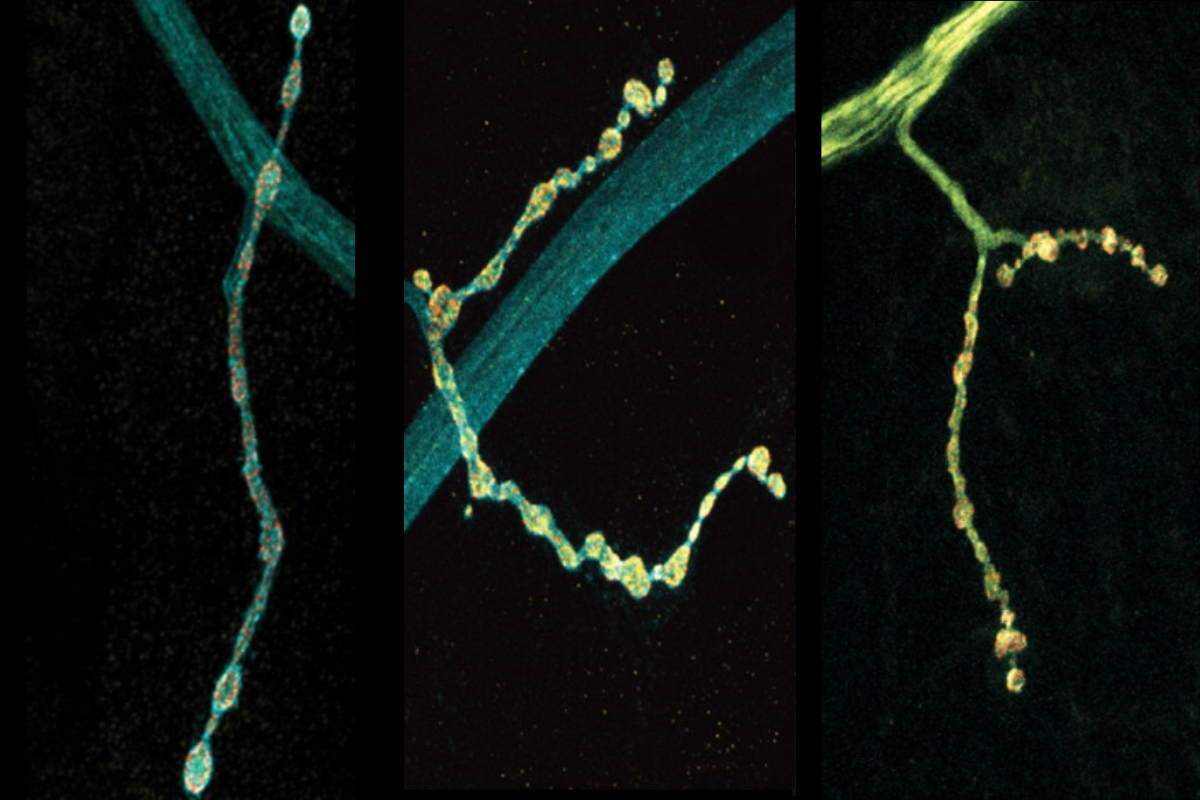
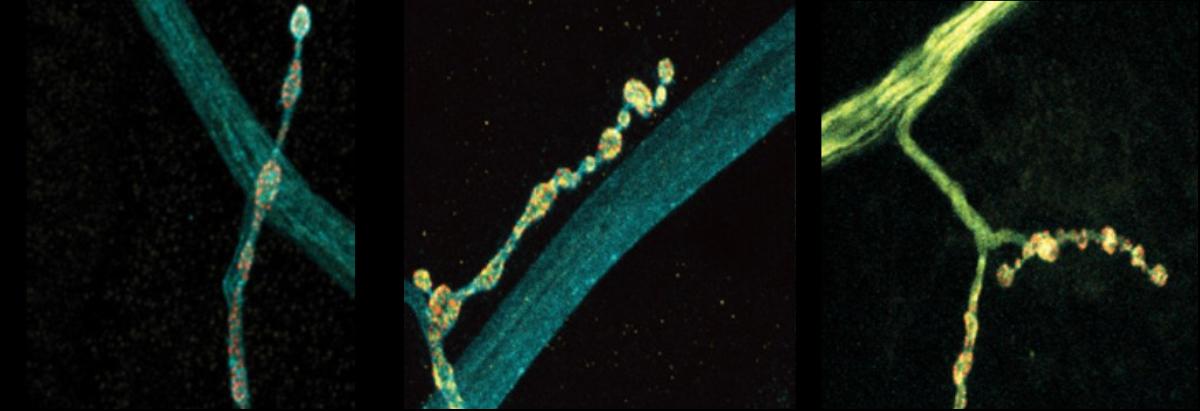
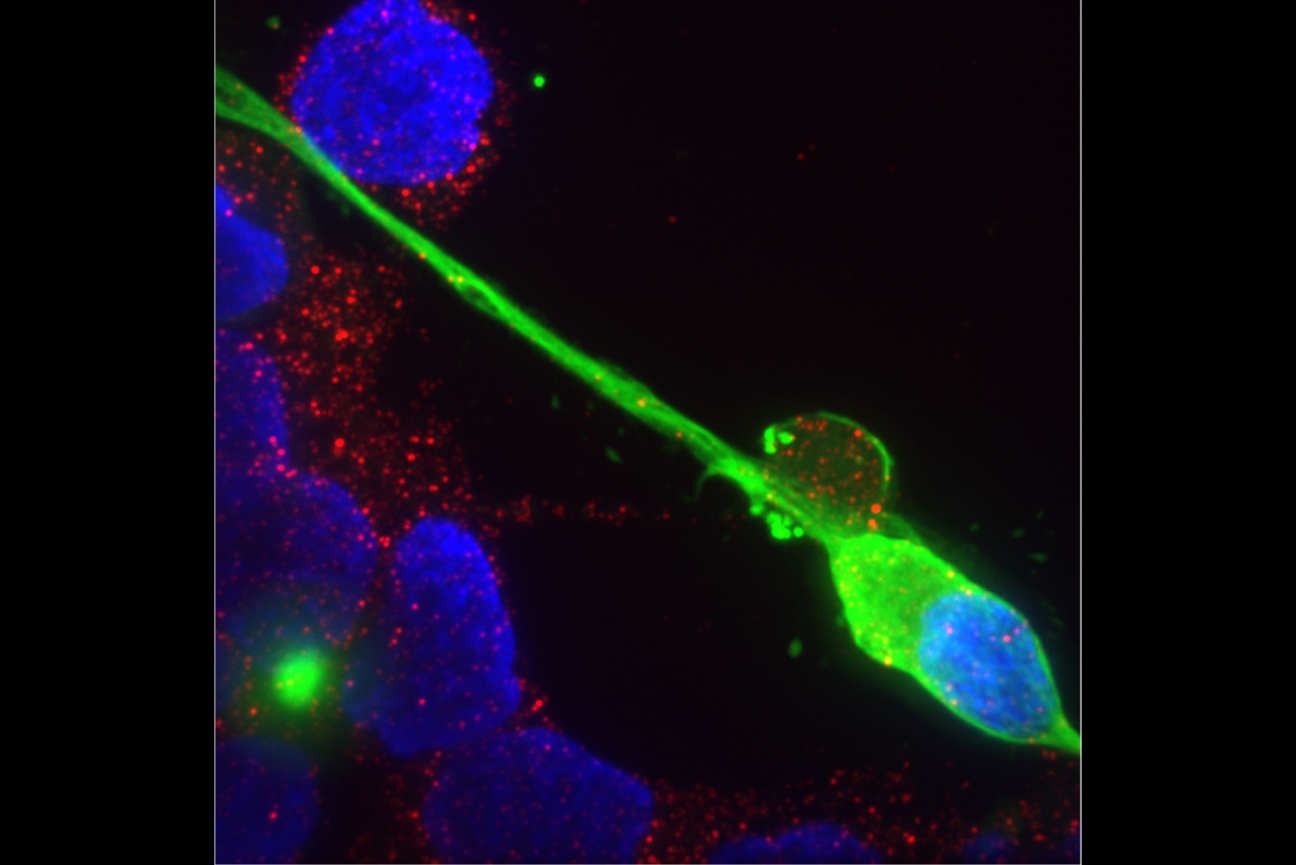
Neurons are talkers. They each communicate with fellow neurons, muscles, or other cells by releasing neurotransmitter chemicals at “synapse” junctions, ultimately producing functions ranging from emotions to motions. But even neurons of the exact same type can vary in their conversational style. A new open-access study in Cell Reports by neurobiologists at The Picower Institute for Learning and Memory highlights a molecular mechanism that might help account for the nuanced diversity of neural discourse.
The scientists made their findings in neurons that control muscles in Drosophila fruit flies. These cells are models in neuroscience because they exhibit many fundamental properties common to neurons in people and other animals, including communication via the release of the neurotransmitter glutamate. In the lab of Troy Littleton, Menicon Professor in MIT’s departments of Biology and Brain and Cognitive Sciences, which studies how neurons regulate this critical process, researchers frequently see that individual neurons vary in their release patterns. Some “talk” more than others.
In more than a decade of studies, Littleton’s lab has shown that a protein called complexin has the job of restraining spontaneous glutamate chatter. It clamps down on fusion of glutamate-filled vesicles at the synaptic membrane to preserve a supply of the neurotransmitter for when the neuron needs it for a functional reason, for instance to simulate a muscle to move. The lab’s studies have identified two different kinds of complexin in flies (mammals have four) and showed that the clamping effectiveness of the rare but potent 7B splice form is regulated by a molecular process called phosphorylation. How the much more abundant 7A version is regulated was not known, but scientists had shown that the RNA transcribed from DNA that instructs the formation of the protein is sometimes edited in the cell by an enzyme called ADAR.
In the new study from Littleton’s team, led by Elizabeth Brija PhD ’23, the lab investigated whether RNA editing of complexin 7A affects how it regulates glutamate release. What she discovered was surprising. Not only does RNA editing of complexin 7A have a significant impact on how well the protein prevents glutamate release, but also this can vary widely among individual neurons because they can stochastically mix and match up to eight different editions of the protein. Some edits were much more common than others on average, but 96 percent of the 200 neurons the team examined had at least some editing, which affected the structure of an end of the protein called its C-terminus. Experiments to test some of the consequences of this structural variation showed that different complexin 7A edits can dramatically affect the level of electrical current measurable at different synapses. That varying level of activity can also affect the growth of the synapses the neurons make with muscle. RNA editing of the protein might therefore endow each neuron with fine degrees of communication control.
“What this offers the nervous system is that you can take the same transcriptome and by alternatively editing various RNA transcripts, these neurons will behave differently,” Littleton says.
Moreover, Littleton and Brija’s team found that other key proteins involved in synaptic glutamate release, such as synapsin and Syx1A, are also sometimes edited at quite different levels among the same population of neurons. This suggests that other aspects of synaptic communication might also be tunable.
“Such a mechanism would be a robust way to change multiple features of neuronal output,” Brija, Littleton, and colleagues wrote.
The team tracked the different editing levels by meticulously extracting and sequencing RNA from the nuclei and cell bodies of 200 motor neurons. The work yielded a rich enough dataset to show that any of three adenosine nucleotides encoding two amino acids in the C-terminus could be swapped for another, yielding eight different editions of the protein. A slim majority of complexin 7A went unedited in the average neuron, while the seven edited versions composed the rest with widely varying degrees of frequency.
To investigate the functional consequences of some of the different editions, the team knocked out complexin and then “rescued” flies by adding back in unedited or two different edited versions. The experiments showed a stark contrast between the two edited proteins. One, which occurs more commonly, proved to be a less effective clamp than unedited complexin, barely preventing spontaneous glutamate release and upticks in electrical current. The other turned out to be more effective at clamping than the unedited version, keeping a tight lid on glutamate release and synaptic output. And while both of the edited versions showed a tendency to drift away from synapses and into the neuron’s axon, the long branch that extends from the cell body, the edition that clamped well prevented any overgrowth of synapses while the one that clamped poorly provided only a meager curb.
Because multiple editions are often present in neurons, Brija and the team did one more set of experiments in which they “rescued” complexin-less flies with a combination of unedited complexin and the weak-clamping edition. The result was a blend of the two: reduced spontaneous glutamate release than with just the weakly clamping edition alone. The findings suggest that not only does each edition potentially fine-tune glutamate release, but that combinations among them can act in a combinatorial fashion.
In addition to Brija and Littleton the paper’s other authors are Zhuo Guan and Suresh Jetti.
The National Institutes of Health, The JPB Foundation, and The Picower Institute for Learning and Memory supported the research.

Sipping a beer on a warm summer evening, one might not consider that humans and yeast have been inextricably linked for thousands of years; winemaking, baking, and brewing all depend on budding yeast. Outside of baking and fermentation, researchers also use Saccharomyces cerevisiae, classified as a fungus, to study fundamental questions of cell biology.
Budding yeast gets its name from the way it multiplies. A daughter cell forms first as a swelling, protruding growth on the mother cell. The daughter cell projects further and further from the mother cell until it detaches as an independent yeast cell.
How do cells decide on a front and back? How do cells decode concentration gradients of chemical signals to orient in useful directions, or sense and navigate around physical obstacles? New Department of Biology faculty member Daniel “Danny” Lew uses the model yeast S. cerevisiae, and a non-model yeast with an unusual pattern of cell division, to explore these questions.
Q: Why is it useful to study yeast, and how do you approach the questions you hope to answer?
A: Humans and yeast are descended from a common ancestor, and some molecular mechanisms developed by that ancestor have been around for so long that yeast and mammals often use the same mechanisms. Many cells develop a front and migrate or grow in a particular direction, like the axons in our nervous system, using similar molecular mechanisms to those of yeast cells orienting growth towards the bud.
When I started my lab, I was working on cell cycle control, but I’ve always been interested in morphogenesis and the cell biology of how cells change shape and decide to do different things with different parts of themselves. Those mechanisms turn out to be conserved between yeast and humans.
But some things are very different about fungal and animal cells. One of the differences is the cell wall and what fungal cells have to do to deal with the fact that they have a cell wall.
Fungi are inflated by turgor pressure, which pushes their membranes against the rigid cell wall. This means they’ll die if there is any hole in the cell wall, which would be expected to happen often as cells remodel the wall in order to grow. We’re interested in understanding how fungi sense when any weak spots appear in the wall and repair them before those weak spots become dangerous.
Yeast cells, like most fungi, also mate by fusing with a partner. To succeed, they must do the most dangerous thing in the fungal lifecycle: get rid of the cell wall at the point of contact to allow fusion. That means they must be precise about where and when they remove the wall. We’re fascinated to understand how they know it is safe to remove the wall there, and nowhere else.
We take an interdisciplinary approach. We’ve used genetics, biochemistry, cell biology, and computational biology to try and solve problems in the past. There’s a natural progression: observation and genetic approaches tend to be the first line of attack when you know nothing about how something works. As you learn more, you need biochemical approaches and, eventually, computational approaches to understand exactly what mechanism you’re looking at.
I’m also passionate about mentoring, and I love working with trainees and getting them fascinated by the same problems that fascinate me. I’m looking to work with curious trainees who love addressing fundamental problems.
Q: How does yeast decide to orient a certain way—towards a mating partner, for example?
A: We are still working on questions of how cells analyze the surrounding environment to pick a direction. Yeast cells have receptors that sense pheromones that a mating partner releases. What is amazing about that is that these cells are incredibly small, and pheromones are released by several potential partners in the neighborhood. That means yeast cells must interpret a very confusing landscape of pheromone concentrations. It’s not apparent how they manage to orient accurately toward a single partner.
That got me interested in related questions. Suppose the cell is oriented toward something that isn’t a mating partner. The cell seems to recognize that there’s an obstacle in the way, and it can change direction to go around that obstacle. This is how fungi get so good at growing into things that look very solid, like wood, and some fungi can even penetrate Kevlar vests.
If they recognize an obstacle, they have to change directions and go around it. If they recognize a mating partner, they have to stick with that direction and allow the cell wall to get degraded. How do they know they’ve hit an obstacle? How do they know a mating partner is different from an obstacle? These are the questions we’d like to understand.
Q: For the last couple of years, you’ve also been studying a budding yeast that forms multiple buds when it reproduces instead of just one. How did you come across it, and what questions are you hoping to explore?
A: I spent several years trying to figure out why most yeasts make one bud and only one bud, which I think is related to the question of why migrating cells make one and only one front. We had what we thought was a persuasive answer to that, so seeing a yeast completely disobey that and make as many buds as it felt like was a shock, which got me intrigued.
We started working on it because my colleague,Amy Gladfelter, had sampled the waters around Woods Hole, Massachusetts. When she saw this specimen under a microscope, she immediately called me and said, “You have to look at this.”
A question we’re very intrigued by is if the cell makes five, seven, or 12 buds simultaneously, how do they divide the mother cell’s material and growth capacity five, seven, or 12 ways? It looks like all of the buds grow at the same rate and reach about the same size. One of our short-term goals is to check whether all the buds really get to exactly the same size or whether they are born unequal.
And we’re interested in more than just growth rate. What about organelles? Do you give each bud the same number of mitochondria, nuclei, peroxisomes, and vacuoles? That question will inevitably lead to follow-up questions. If each bud has the same number of mitochondria, how does the cell measure mitochondrial inheritance to do that? If they don’t have the same amount, then buds are each born with a different complement and ratio of organelles. What happens to buds if they have very different numbers of organelles?
As far as we can tell, every bud gets at least one nucleus. How the cell ensures that each bud gets a nucleus is a question we’d also very much like to understand.
We have molecular candidates because we know a lot about how model yeasts deliver nuclei, organelles, and growth materials from the mother to the single bud. We can mutate candidate genes and see if similar molecular pathways are involved in the multi-budding yeast and, if so, how they are working.
It turns out that this unconventional yeast has yet to be studied from the point of view of basic cell biology. The other thing that intrigues me is that it’s a poly-extremophile. This yeast can survive under many rather harsh conditions: it’s been isolated in Antarctica, from jet engines, from all kinds of plants, and of course from the ocean as well. An advantage of working with something so ubiquitous is we already know it’s not toxic to us under almost any circumstances. We come into contact with it all the time. If we learn enough about its cell biology to begin to manipulate it, then there are many potential applications, from human health to agriculture.
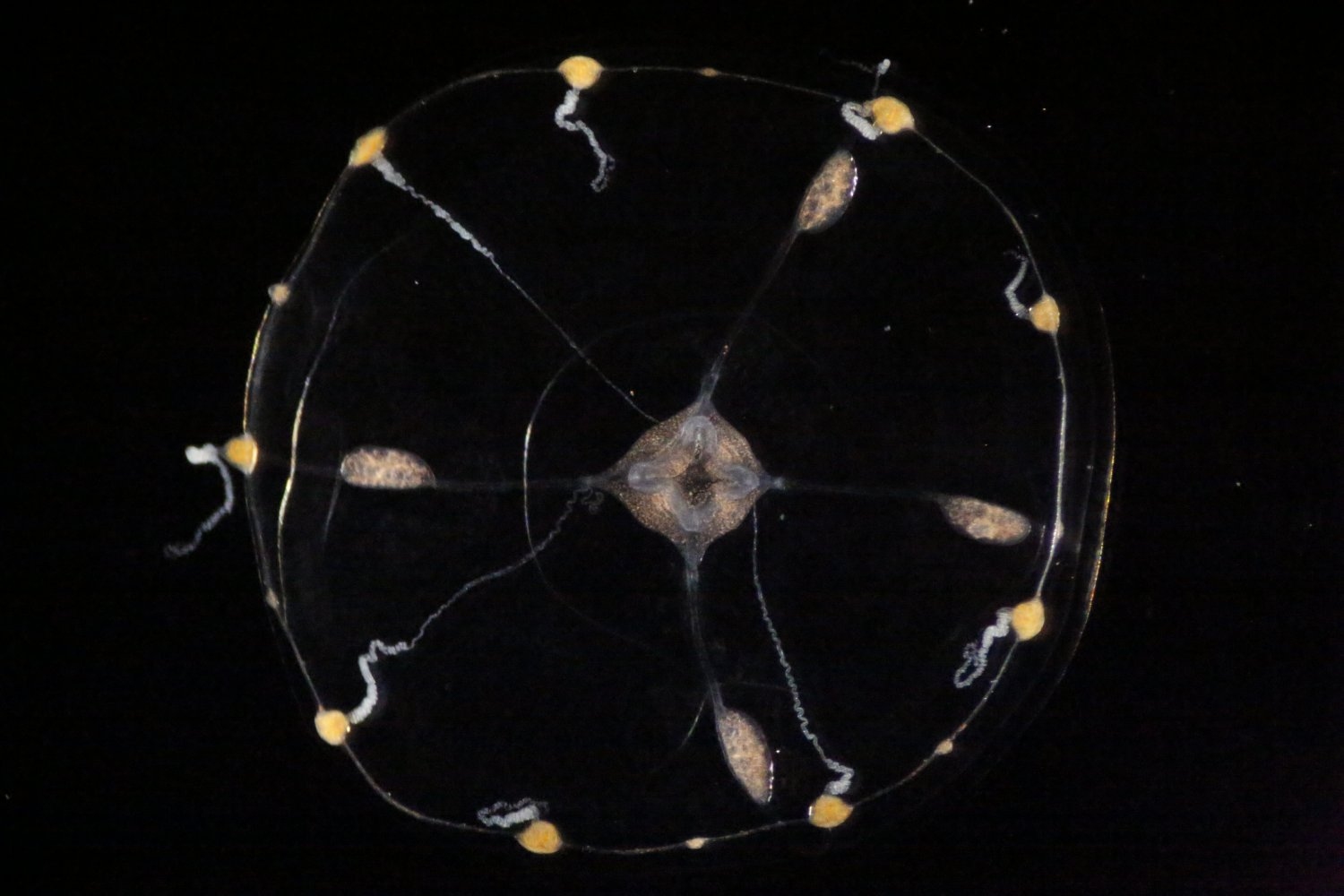
The Clytia hemisphaerica jellyfish is not only a hypnotically graceful swimmer, but also an amazing neuron-manufacturing machine with a remarkable ability to expand and regenerate its nervous system.
Now, thanks to a prestigious Klingenstein-Simons Fellowship Award in Neuroscience, MIT Assistant Professor Brady Weissbourd will study how the tiny, transparent animals use this ability to build, organize, and rebuild a stable, functional, and robust nervous system throughout their lives.
“As we look more broadly across the animal kingdom it is amazing to see how similar the basic biology is of animals that look completely different — even jellyfish have neurons similar to our own that generate their behavior,” says Weissbourd, a faculty member in MIT’s Department of Biology and The Picower Institute for Learning and Memory, whose work to engineer genetic access to C. hemisphaerica in 2021 established it as a new neuroscience model organism. “At the same time, it could be just as important to examine what is different across species, particularly when it comes to some of the incredible capabilities that have evolved.”
Weissbourd is just one of 13 researchers nationally to be recognized with this fellowship, which provides $300,000 over three years. It will enable Weissbourd’s lab to tackle several questions raised by the jellyfish’s prodigious production of neurons. Where does the constant stream of newborn neurons come from, and what guides them to their eventual places in the jellyfish’s mesh-like neural network? How does the jellyfish organize these ever-changing neural populations — for instance, into functional circuits — to enable its various behaviors?
Another question hails from the surprising results of an experiment in which Weissbourd ablated the entire class of the neurons that the jellyfish uses to fold up its umbrella-shaped body — about 10 percent of the 10,000 or so neurons that it has. He found that within a week enough new neurons had taken their place that the folding behavior was restored. Weissbourd’s studies will also seek to determine how the animal can so readily bounce back from the destruction of a whole major neural network and the behavior it produces.
“We were studying the neural control of a particular behavior and stumbled across this shocking observation that the subnetwork that controls this behavior is constantly changing size and can completely regenerate,” Weissbourd says. “We want to understand the mechanisms that allow this network to be so robust, including the ability to rebuild itself from scratch. I’m very grateful to the Klingenstein Fund and the Simons Foundation for supporting our work.”