Eva Frederick | Whitehead Institute
August 31, 2022
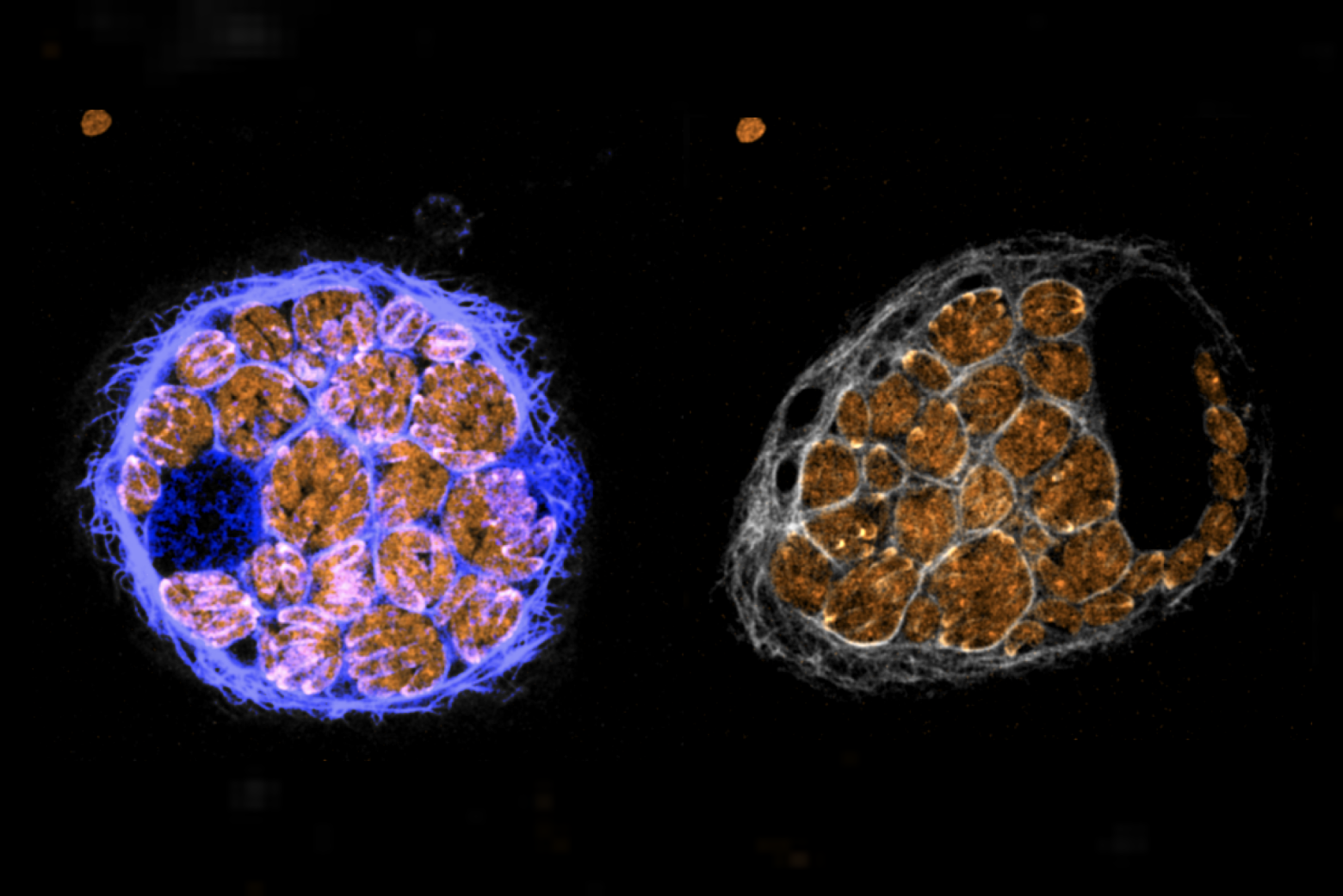
Much like humans, plants, and bacteria, the single-celled parasite Toxoplasma gondii (T. gondii) uses calcium as a messenger to coordinate important cellular processes. But while the messenger is the same, the communication pathways that form around calcium differ significantly between organisms.
“Since Toxoplasma parasites are so divergent from us, they have evolved their own sets of proteins that are involved in calcium signaling pathways,” said Alice Herneisen, a graduate student in the lab of Whitehead Institute Member Sebastian Lourido.
Lourido and his lab study the molecular mechanisms that allow the single-celled parasite T. gondii and related pathogens to be so widespread and potentially deadly — and calcium signaling is an important part of the parasite’s process of invading its hosts. “Calcium governs this very important transition from the parasites replicating inside of host cells to parasites leaving those cells and seeking out new ones to infect,” said Lourido. “We’ve been really interested in how calcium plays into the regulation of proteins inside the parasite.”
A paper published August 17 in eLife provides some insight. In the paper, Herneisen, Lourido and collaborators used an approach called thermal profiling to broadly survey which parasite proteins are involved in calcium signaling in T. gondii. The new work reveals that an unexpected protein plays a role in parasite calcium pathways, and provides new targets that scientists could potentially use to stop the spread of the parasite. The data will also serve as a resource that other Toxoplasma researchers can use to find out whether their own proteins of interest interact with calcium pathways in parasite cells.
The heat is on
When studying calcium pathways in humans, researchers can often draw parallels from work in mice. “But parasites are very different from us,” said Lourido. “All of the principles that we’ve learned about calcium signaling in humans or mice can’t be readily translated over to parasites.”
So to study these mechanisms in Toxoplasma, the researchers had to start from scratch to determine which proteins were involved. That’s where the thermal profiling method came in. The method is based on the observation that proteins are designed to work well at specific temperatures, and when it becomes too hot for them, they melt. Consider eggs: when the proteins in egg whites and egg yolks are heated in a frying pan, the proteins begin to melt and congeal. “When we think about a protein melting, what we mean is the proteins unraveling,” said Lourido. “When proteins unravel, they expose side chains that bind to each other. They stop being individual proteins that are well-folded, and become a mesh.”
The thermal profiling method works by applying heat to parasite cells and graphing how each of the parasite’s proteins responds to changes in temperature under different conditions (for example, the presence or absence of calcium). In a 2020 paper, the researchers used the thermal profiling method to investigate the role of a protein called ENH1 in calcium signaling.
In their new paper, Lourido and Herneisen investigated the effect of calcium on all proteins in the parasite using two approaches. The researchers combined parasites with specific amounts of calcium, applied heat, and then performed proteomics techniques to track how the calcium affected the melting behavior of each protein. If a protein’s melting point was higher or lower than usual, the researchers could deduce that that protein was changed either by calcium itself or by another player in a calcium signaling pathway.
They then treated the parasites with a chemical that caused them to release stored calcium in a controlled manner and measured how a protein modification called phosphorylation changed over time. Together, these methods allowed them to infer how proteins might sense and respond to calcium within the signaling network.
Their approach provided data on nearly every expressed protein in the parasite cells, but the researchers zeroed in on one particular protein called Protein Phosphatase 1 (or PP1). The protein is ubiquitous across many species, but has never previously been implicated in calcium signaling pathways. They found that the protein was concentrated at the front end of the parasite. This region of the parasite cell is involved in motility and host invasion.
The protein’s role in the parasites — and in the other organisms in which it appears — is to remove the small molecules called phosphates from phosphorylated proteins. “This is a modification that can often change the activity of individual proteins, because it’s this big charge that’s been covalently stuck onto the surface of the protein,” Lourido said. “This ends up being a principle through which many, many different biological processes are regulated.”
How exactly PP1 interacts with calcium remains to be seen. When the researchers depleted PP1 in parasite cells, they found that the protein is somehow involved in helping the parasite take in calcium necessary for movement. It’s unclear whether or not it actually binds calcium or is involved in the pathway through another mechanism.
Because parasites use calcium signaling to coordinate life cycle changes such as entering or leaving host cells, insights into the key players in calcium pathways could be a boon to public health. “These are kind of the pressure points or the hubs that would be ideal to target in order to prevent the spread and pathogenesis of these parasites,” Herneisen said.
Herneisen and collaborators focused primarily on PP1, but there are many other proteins to investigate using the data from this project. “I think part of the reason why I wanted to release this paper is so that the field could take the next steps,” she said. “I’m just one person — it would be great if 20 other people find that the protein that they were studying is calcium responsive, and they can chase down the exact reason for that or how it is involved in this greater calcium signaling network. This was exciting for us with regards to PP1, and I’m sure other researchers will make their own connections.”
Notes
Alice L. Herneisen, Zhu-Hong Li, Alex W. Chan, Silvia NJ Moreno, and Sebastian Lourido. “Temporal and thermal profiling of the Toxoplasma proteome implicates parasite Protein Phosphatase 1 in the regulation of Ca2+-responsive pathways”. eLife, August 17, 2022. DOI: https://doi.org/10.7554/eLife.80336
