Two proteins found to induce cell death through incomplete base excision repair
Depletion of either the DapB or Dxr proteins causes oxidative stress and cell death in bacteria, which could aid the development of more effective antibiotics.
Grace van Deelen
How do bacteria die? It’s an important question, especially since these single-celled organisms seem to be outpacing the development of new antibiotics. However, any one bacterial cell will often die from a number of separate but related pathways acting simultaneously, making understanding bacterial death difficult. Determining how to induce those pathways — and the role each pathway plays in a cell’s death — is key to creating effective antibiotics, especially after bacteria evolve to resist some drugs. But new research from Graham Walker’s lab in the MIT Department of Biology suggests that one historically under-appreciated cause of bacterial death, called oxidative stress, could help scientists develop antibiotics that kill bacteria more effectively.
Many antibiotics target the bacteria’s cell wall or replication process. However, some antibiotics can additionally cause changes in a cell’s metabolism that lead to a phenomenon called oxidative stress. Reactive oxygen-containing molecules float around freely inside the cell, sometimes bumping into other molecules, reacting with them, and stealing their unpaired electrons in a process called oxidation. For example, a guanine molecule — the DNA nucleotide commonly abbreviated to “G” — may become an oxidized guanine called 8-oxo-dG, a transformation that causes mutations in a cell’s genetic code. The harmful effects of oxidation are usually managed by the cell, but the disruption caused by these antibiotics can also become fatal to the cell.
In the case of 8-oxo-dG, the cell responds to this oxidative stress by attempting to cut the oxidized guanine out of the genome and repair it with a regular nucleotide during a process called base excision repair (BER). However, during BER, every completed step produces intermediate substances, including other forms of damaged DNA, which then must be cleared by another enzyme or protein. But sometimes the cell is unable to complete BER because these intermediate substances build up. When there is an imbalance of intermediate substances, the cell pauses the repair, leaving breaks in the strands of DNA that cause cell death.
Because incomplete BER is just one of many contributing causes of cell death, the total contribution of incomplete BER to cell death remained unclear. As a result, scientists in the Walker lab were interested in determining other stressors, besides known antibiotics, that might cause incomplete BER of 8-oxo-dG. “If this mechanism of 8-oxo-dG getting into DNA causes bacteria to die, there’s probably some other stressor that isn’t an antibiotic that would cause cells to die by the same way,” says Walker.
In the paper, published on February 8 in mBio, the researchers determined two additional stressors that also induce cell death via incomplete BER of 8-oxo-dG. They found that the depletion of proteins DapB and Dxr also induced oxidative stress and incomplete BER of 8-oxo-dG. Scientists have known of these proteins — both of which are involved in bacterial metabolism — for some time, but had never associated them with incomplete BER.
“Incomplete base excision repair is probably one of more underappreciated ways a cell can die,” Walker says. “So we wanted to explore that pathway further.”
Charley Gruber, a postdoc in the Walker lab and lead author on the paper, identified DapB and Dxr by screening a library of 238 proteins essential for Escherichia coli growth. He determined that, in the absence of these two proteins, the cell overproduced the reactive oxygen-containing molecules that contribute to oxidative stress. As a result, the oxidized nucleotide 8-oxo-dG was incorporated into the genome, leading to cell death through incomplete BER. Researchers don’t know for sure why depletion of DapB and Dxr increases the amount of reactive oxygen-containing molecules inside the cell, but oxidative stress is a common reaction to many disruptions that bacterial cells may face.
To Walker and Gruber’s surprise, their results also showed that the total contribution of incomplete BER to cell death was different between the two proteins — Dxr-depleted cells died faster than DapB-depleted cells, suggesting that a lack of Dxr played a larger role in cell death. Because the responses to protein depletion were so different between DapB and Dxr, the researchers concluded that there is no singular pathway that causes oxidative stress; rather, it is probably a common consequence of many possible disruptions to bacterial cell physiology.
“If there’s one important thing I think we need to realize about cell death,” Gruber says, “it’s that a lot is happening to a stressed cell. And what is actually lethal might differ between two cells.”
This study adds to a body of research by Gruber, Walker, and others about the role of incomplete BER in the process of cell death. In 2012, the Walker lab published a paper in Science — building on earlier work from MIT’s Termeer Professor of Bioengineering, Jim Collins — which showed for the first time that some commonly-used antibiotics kill by way of oxidative stress and the 8-oxo-dG pathway of incomplete BER. The idea was not immediately accepted by the scientific community, and a debate ensued: Shortly after Walker’s paper, Northeastern University biologist Kim Lewis and University of Illinois biologist Jim Imlay each published separate papers suggesting that bactericidal antibiotics had nothing to do with oxidative stress. Since then, the Walker and Collins labs have continued to research the topic, producing more supporting data for their argument that oxidative stress and incomplete BER are, in fact, an important pathway of cell death.
“This new work provides a strong genetic foundation for the role of incomplete BER in bacterial cell death,” Collins says . “Oxidative stress and BER should be targeted as a means to potentiate existing antibiotics and enhance our antibiotic arsenal.”
Scientific debates like the one surrounding the contribution of incomplete BER to bacterial death are crucial to the creation of effective antibiotics. Most antibiotics work by breaking the cell wall and causing cell death that way. However, the lab’s findings offer a possibility for antibiotic assistance: the common practice of using secondary antibiotics to aid in cell death thorough a different pathway. For example, administering a secondary antibiotic that triggers the 8-oxo-dG pathway along with the primary antibiotic that is lethal to bacteria through cell wall destruction could be more effective than one antibiotic on its own, Gruber suggests.
“Many of our antibiotics are not working, or we’ve overused them in some cases, so we’re really running out of drugs,” he says. “So an antibiotic that induces oxidative stress could be another way to help existing drugs work better.”
—
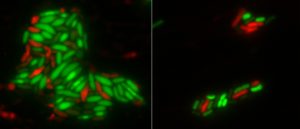
Top image: E. coli cells with either DapB (left) or Dxr (right) depleted. Living cells are stained green while dead cells are stained red. Credit: Charley Gruber
Citation:
“Degradation of the Escherichia coli Essential Proteins DapB and Dxr Results in Oxidative Stress, which Contributes to Lethality through Incomplete Base Excision Repair”
mBio, online February 8, 2022, DOI: 10.1128/mbio.03756-21
Charley C. Gruber, Vignesh M. P. Babu, Kamren Livingston, Heer Joisher, and Graham C. Walker
