Researchers discover a new toxin that impedes bacterial growth
Raleigh McElvery
An international research collaboration has discovered a new toxin, which bacteria inject into their neighboring cells to hinder growth and compete for limited resources. Their findings were published on November 6 in Nature.
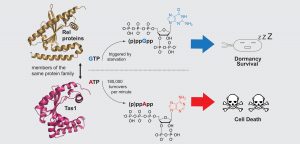
At McMaster University in Ontario, Canada, co-senior author John Whitney and his team were studying a secretion system that allows bacteria to deliver these deleterious molecules, when they came across a new toxin. This toxin was an enzyme, and one they had never seen before. Based on their structural analyses, it looked a lot like the enzymes that synthesize guanosine tetra- and penta-phosphate, collectively known as “(p)ppGpp.” (p)ppGpp is a signaling molecule that helps bacteria safely dial down their growth rate in response to starvation. Suspecting the toxin might produce (p)ppGpp in recipient cells and ultimately impact their growth, the McMaster team shared their findings with Michael Laub, a professor of biology at MIT and a Howard Hughes Medical Institute investigator.
Boyuan Wang, a postdoc in the Laub lab who specializes in (p)ppGpp synthesis, examined the unknown enzyme’s activity to determine its product. He soon realized that, rather than making (p)ppGpp, this enzyme was instead producing related molecules, adenosine tetraphosphate and adenosine pentaphosphate, collectively referred to as (p)ppApp. Somehow, (p)ppApp production was hindering growth.
“Scientists have known about (p)ppApp for decades, but it hadn’t been shown to have a physiological role in organisms until now,” says Wang, a co-first author. Researchers had previously speculated that (p)ppApp was merely a non-specific product generated during (p)ppGpp synthesis, so it was surprising to find an enzyme that made it specifically.
The researchers named their enzyme Tas1, and determined that it uses the cell’s main energy currency, ATP, and its precursor, ADP, to produce (p)ppApp. In fact, one molecule of Tas1 was enough to consume 180,000 molecules of ATP per minute — two orders of magnitude faster than the fastest known (p)ppGpp synthetases work to make (p)ppGpp. Using metabolomic analyses, the MIT group showed that this exceptional rate of (p)ppApp production requires so much energy that there’s not enough left to carry out essential cellular processes, effectively killing the bacterium.
“Bacteria can inject only one Tas1 molecule at a time, and yet the toxin has such a powerful impact on its target, depleting the ATP supply in a matter of minutes,” Wang says. “The secretion system is kind of like a miniaturized intercontinental ballistic missile in terms of its structure and impact, except it functions ‘intercompartmentally’ between two bacteria.”
“It’s amazing that the first (p)ppApp synthase ever discovered actually serves as a novel, and quite clever, means of killing another cell,” says Laub, a co-senior author. “Findings like these really highlight the diversity of mechanisms that bacteria use to inhibit each other’s growth.”
Tas1, the researchers believe, may augment other known toxins that bacteria inject into one another to hinder cell growth, including those that work in the cytoplasm or target the cell envelope.
As a biochemist, Wang is excited by the prospect of using Tas1 as a tool in future experiments to deplete ATP, and probe the networks of metabolic regulation within bacteria and higher organisms.
“It’s fascinating to uncover the strategies nature uses to repurpose proteins,” Wang says. “Before this study, we wouldn’t have considered the possibility that a member of this protein family could be used as a deadly toxin.”
Image: Tas1, a newly discovered enzyme, has a similar structure to the widespread bacterial Rel proteins that produce (p)ppGpp to promote survival during starvation. Tas1 alters its specificity to quickly produce large amounts of (p)ppApp, serving as a toxin in Pseudomonas aeruginosa and killing competing bacteria. Credit: Boyuan Wang.
Citation:
“An interbacterial toxin inhibits target cell growth by synthesizing (p)ppApp”
Nature, online November 6, 2019, DOI: 10.1038/s41586-019-1735-9
Shehryar Ahmad, Boyuan Wang, Matthew D. Walker, Hiu-Ki R. Tran, Peter J. Stogios, Alexei Savchenko, Robert A. Grant, Andrew G. McArthur, Michael T. Laub, and John C. Whitney
