

There are many hurdles to clear before a research discovery becomes a life-changing treatment for patients. That’s especially true when the treatments being developed represent an entirely new class of medicines. But overcoming those obstacles can revolutionize our ability to treat diseases.
Few companies exemplify that process better than Alnylam Pharmaceuticals. Alnylam was founded by a group of MIT-affiliated researchers who believed in the promise of a technology — RNA interference, or RNAi.
The researchers had done foundational work to understand how RNAi, which is a naturally occurring process, works to silence genes through the degradation of messenger RNA. But it was their decision to found Alnylam in 2002 that attracted the funding and expertise necessary to turn their discoveries into a new class of medicines. Since that decision, Alnylam has made remarkable progress taking RNAi from an interesting scientific discovery to an impactful new treatment pathway.
Today Alnylam has five medicines approved by the U.S. Food and Drug Administration (one Alnylam-discovered RNAi therapeutic is licensed to Novartis) and a rapidly expanding clinical pipeline. The company’s approved medicines are for debilitating, sometimes fatal conditions that many patients have grappled with for decades with few other options.
The company estimates its treatments helped more than 5,000 patients in 2023 alone. Behind that number are patient stories that illustrate how Alnylam has changed lives. A mother of three says Alnylam’s treatments helped her take back control of her life after being bed-ridden with attacks associated with the rare genetic disease acute intermittent porphyria (AIP). Another patient reported that one of the company’s treatments helped her attend her daughter’s wedding. A third patient, who had left college due to frequent AIP attacks, was able to return to school.
These days Alnylam is not the only company developing RNAi-based medicines. But it is still a pioneer in the field, and the company’s founders — MIT Institute Professor Phil Sharp, Professor David Bartel, Professor Emeritus Paul Schimmel, and former MIT postdocs Thomas Tuschl and Phillip Zamore — see Alnylam as a champion for the field more broadly.
“Alnylam has published more than 250 scientific papers over 20 years,” says Sharp, who currently serves as chair of Alnylam’s scientific advisory board. “Not only did we do the science, not only did we translate it to benefit patients, but we also described every step. We established this as a modality to treat patients, and I’m very proud of that record.”
Pioneering RNAi development
MIT’s involvement in RNAi dates back to its discovery. Before Andrew Fire PhD ’83 shared a Nobel Prize for the discovery of RNAi in 1998, he worked on understanding how DNA was transcribed into RNA, as a graduate student in Sharp’s lab.
After leaving MIT, Fire and collaborators showed that double-stranded RNA could be used to silence specific genes in worms. But the biochemical mechanisms that allowed double-stranded RNA to work were unknown until MIT professors Sharp, Bartel, and Ruth Lehmann, along with Zamore and Tuschl, published foundational papers explaining the process. The researchers developed a system for studying RNAi and showed how RNAi can be controlled using different genetic sequences. Soon after Tuschl left MIT, he showed that a similar process could also be used to silence specific genes in human cells, opening up a new frontier in studying genes and ultimately treating diseases.
“Tom showed you could synthesize these small RNAs, transfect them into cells, and get a very specific knockdown of the gene that corresponded to that the small RNAs,” Bartel explains. “That discovery transformed biological research. The ability to specifically knockdown a mammalian gene was huge. You could suddenly study the function of any gene you were interested in by knocking it down and seeing what happens. … The research community immediately started using that approach to study the function of their favorite genes in mammalian cells.”
Beyond illuminating gene function, another application came to mind.
“Because almost all diseases are related to genes, could we take these small RNAs and silence genes to treat patients?” Sharp remembers wondering.
To answer the question, the researchers founded Alnylam in 2002. (They recruited Schimmel, a biotech veteran, around the same time.) But there was a lot of work to be done before the technology could be tried in patients. The main challenge was getting RNAi into the cytoplasm of the patients’ cells.
“Through work in Dave Bartel and Phil Sharp’s lab, among others, it became evident that to make RNAi into therapies, there were three problems to solve: delivery, delivery, and delivery,” says Alnylam Chief Scientific Officer Kevin Fitzgerald, who has been with the company since 2005.
Early on, Alnylam collaborated with MIT drug delivery expert and Institute Professor Bob Langer. Eventually, Alnylam developed the first lipid nanoparticles (LNPs) that could be used to encase RNA and deliver it into patient cells. LNPs were later used in the mRNA vaccines for Covid-19.
“Alnylam has invested over 20 years and more than $4 billion in RNAi to develop these new therapeutics,” Sharp says. “That is the means by which innovations can be translated to the benefit of society.”
From scientific breakthrough to patient bedside
Alnylam received its first FDA approval in 2018 for treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis, a rare and fatal disease. It doubled as the first RNAi therapeutic to reach the market and the first drug approved to treat that condition in the United States.
“What I keep in mind is, at the end of the day for certain patients, two months is everything,” Fitzgerald says. “The diseases that we’re trying to treat progress month by month, day by day, and patients can get to a point where nothing is helping them. If you can move their disease by a stage, that’s huge.”
Since that first treatment, Alnylam has updated its RNAi delivery system — including by conjugating small interfering RNAs to molecules that help them gain entry to cells — and earned approvals to treat other rare genetic diseases along with high cholesterol (the treatment licensed to Novartis). All of those treatments primarily work by silencing genes that encode for the production of proteins in the liver, which has proven to be the easiest place to deliver RNAi molecules. But Alnylam’s team is confident they can deliver RNAi to other areas of the body, which would unlock a new world of treatment possibilities. The company has reported promising early results in the central nervous system and says a phase one study last year was the first RNAi therapeutic to demonstrate gene silencing in the human brain.
“There’s a lot of work being done at Alnylam and other companies to deliver these RNAis to other tissues: muscles, immune cells, lung cells, etc.,” Sharp says. “But to me the most interesting application is delivery to the brain. We think we have a therapeutic modality that can very specifically control the activity of certain genes in the nervous system. I think that’s extraordinarily important, for diseases from Alzheimer’s to schizophrenia and depression.”
The central nervous system work is particularly significant for Fitzgerald, who watched his father struggle with Parkinson’s.
“Our goal is to be in every organ in the human body, and then combinations of organs, and then combinations of targets within individual organs, and then combinations of targets within multi-organs,” Fitzgerald says. “We’re really at the very beginning of what this technology is going do for human health.”
It’s an exciting time for the RNAi scientific community, including many who continue to study it at MIT. Still, Alnylam will need to continue executing in its drug development efforts to deliver on that promise and help an expanding pool of patients.
“I think this is a real frontier,” Sharp says. “There’s major therapeutic need, and I think this technology could have a huge impact. But we have to prove it. That’s why Alnylam exists: to pursue new science that unlocks new possibilities and discover if they can be made to work. That, of course, also why MIT is here: to improve lives.”
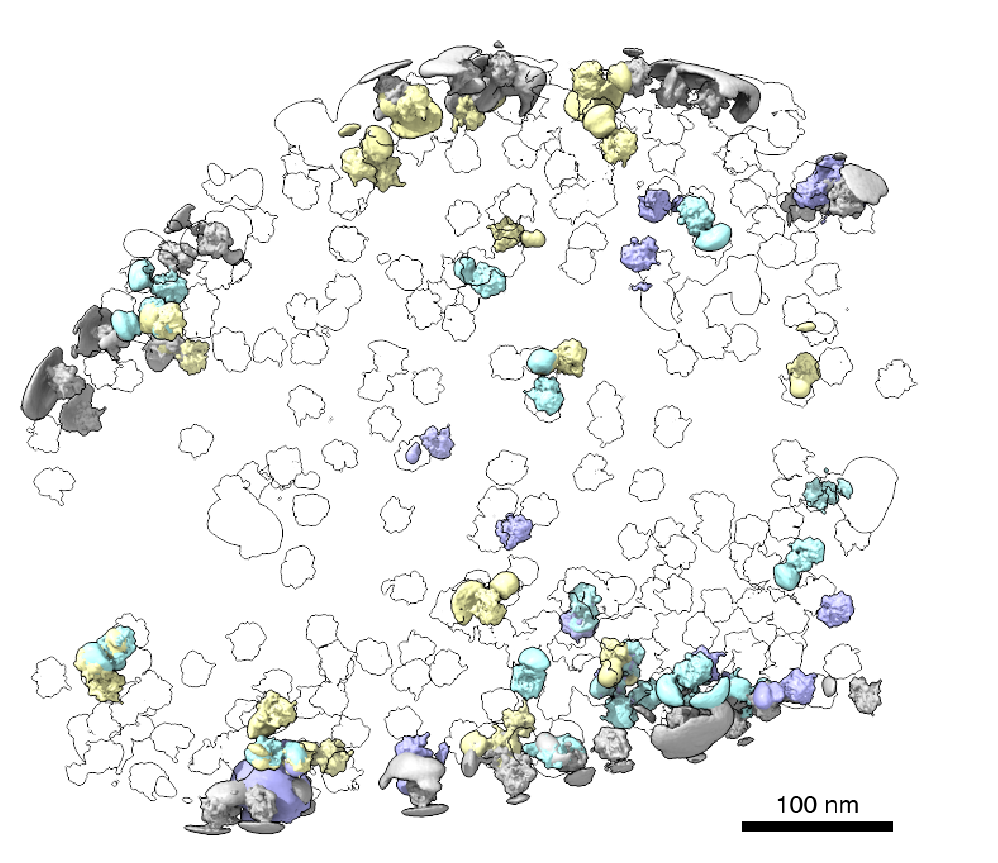
Cells rely on complex molecular machines composed of protein assemblies to perform essential functions such as energy production, gene expression, and protein synthesis. To better understand how these machines work, scientists capture snapshots of them by isolating proteins from cells and using various methods to determine their structures. However, isolating proteins from cells also removes them from the context of their native environment, including protein interaction partners and cellular location.
Recently, cryogenic electron tomography (cryo-ET) has emerged as a way to observe proteins in their native environment by imaging frozen cells at different angles to obtain three-dimensional structural information. This approach is exciting because it allows researchers to directly observe how and where proteins associate with each other, revealing the cellular neighborhood of those interactions within the cell.
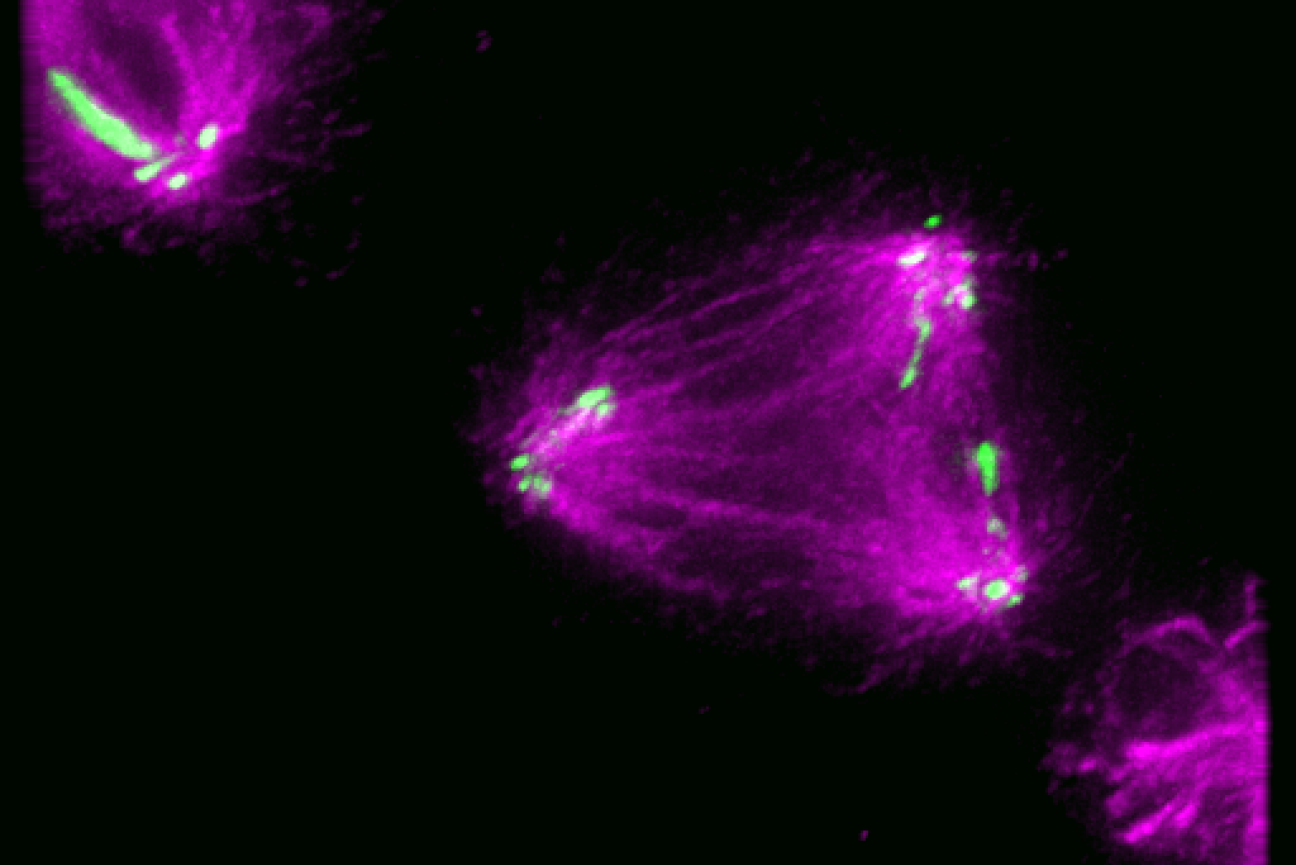
With the technology available to image proteins in their native environment, graduate student Barrett Powell wondered if he could take it one step further: what if molecular machines could be observed in action? In a paper published today in Nature Methods, Powell describes the method he developed, called tomoDRGN, for modeling structural differences of proteins in cryo-ET data that arise from protein motions or proteins binding to different interaction partners. These variations are known as structural heterogeneity.
Although Powell had joined the Davis Lab as an experimental scientist, he recognized the potential impact of computational approaches in understanding structural heterogeneity within a cell. Previously, the Davis Lab developed a related methodology named cryoDRGN to understand structural heterogeneity in purified samples. As Powell and Associate Professor of Biology Joey Davis saw cryo-ET rising in prominence in the field, Powell took on the challenge of reimagining this framework to work in cells.
When solving structures with purified samples, each particle is imaged only once. By contrast, cryo-ET data is collected by imaging each particle more than 40 times from different angles. That meant tomoDRGN needed to be able to merge the information from more than 40 images, which was where the project hit a roadblock: the amount of data led to an information overload.
To address the information overload, Powell successfully rebuilt the cryoDRGN model to prioritize only the highest-quality data. When imaging the same particle multiple times, radiation damage occurs. The images acquired earlier, therefore, tend to be of higher quality because the particles are less damaged.
“By excluding some of the lower quality data, the results were actually better than using all of the data–and the computational performance was substantially faster,” Powell says.
Just as Powell was beginning work on testing his model, he had a stroke of luck: the authors of a groundbreaking new study that visualized, for the first time, ribosomes inside cells at near-atomic resolution, shared their raw data on the Electric Microscopy Public Image Archive (EMPIAR). This dataset was an exemplary test case for Powell, through which he demonstrated that tomoDRGN could uncover structural heterogeneity within cryo-ET data.
According to Powell, one exciting result is what tomoDRGN found surrounding a subset of ribosomes in the EMPIAR dataset. Some of the ribosomal particles were associated with a bacterial cell membrane and engaged in a process called cotranslational translocation. This occurs when a protein is being simultaneously synthesized and transported across a membrane. Researchers can use this result to make new hypotheses about how the ribosome functions with other protein machinery integral to transporting proteins outside of the cell, now guided by a structure of the complex in its native environment.
After seeing that tomoDRGN could resolve structural heterogeneity from a structurally diverse dataset, Powell was curious: how small of a population could tomoDRGN identify? For that test, he chose a protein named apoferritin which is a commonly used benchmark for cryo-ET and is often treated as structurally homogeneous. Ferritin is a protein used for iron storage and is referred to as apoferritin when it lacks iron.
Surprisingly, in addition to the expected particles, tomoDRGN revealed a minor population of ferritin particles–with iron bound–making up just 2% of the dataset that was not previously reported. This result further demonstrated tomoDRGN’s ability to identify structural states that occur so infrequently that they would be averaged out with traditional analysis tools.
Powell and other members of the Davis Lab are excited to see how tomoDRGN can be applied to further ribosomal studies and to other systems. Davis works on understanding how cells assemble, regulate, and degrade molecular machines, so the next steps include exploring ribosome biogenesis within cells in greater detail using this new tool.
“What are the possible states that we may be losing during purification?” Davis says. “Perhaps more excitingly, we can look at how they localize within the cell and what partners and protein complexes they may be interacting with.”
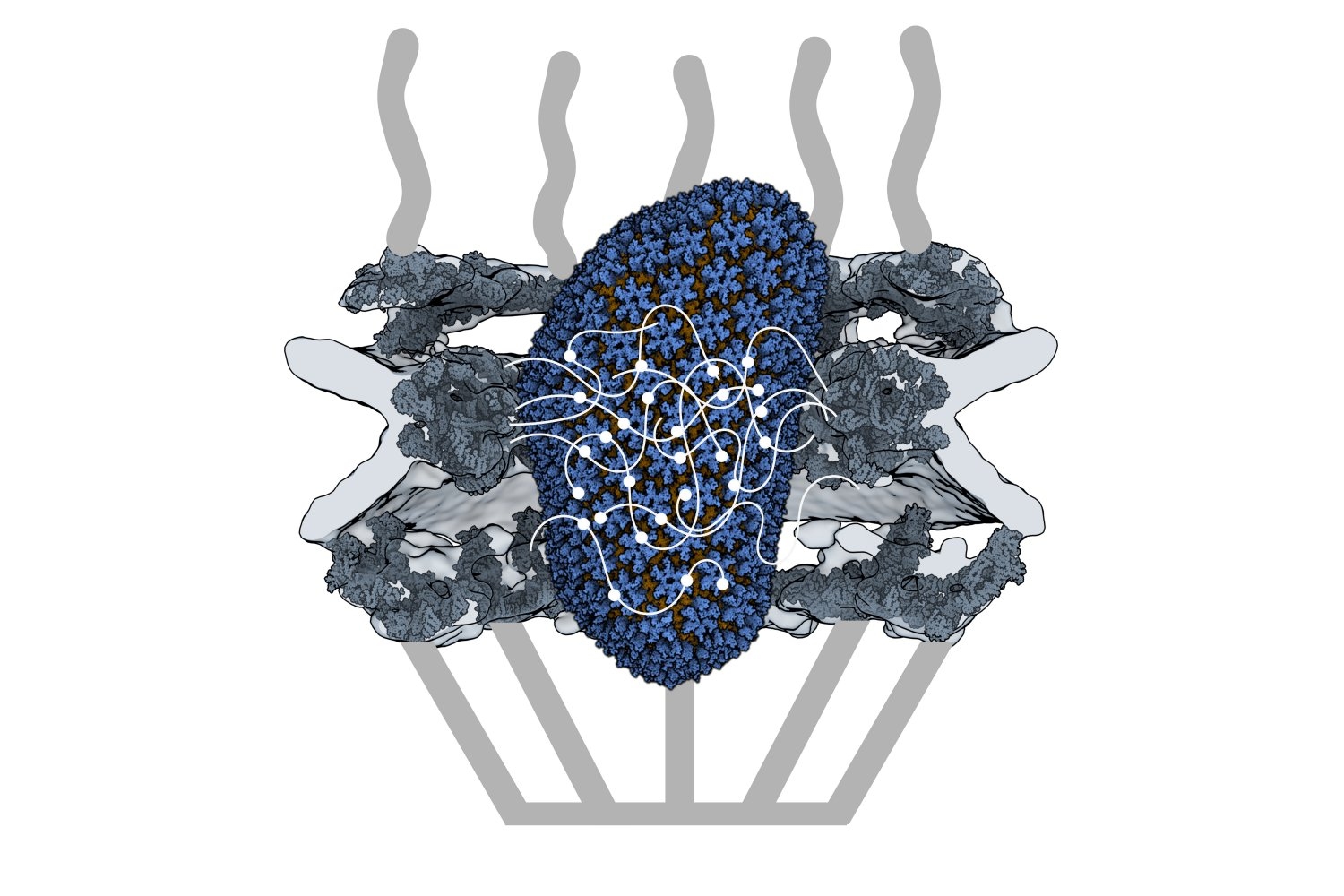
Retroviruses cannot replicate on their own — they must insert their genetic code into the DNA of a host and exploit the host cell’s resources to make more copies of themselves, furthering infection. Some retroviruses only infect cells as they divide, when the nuclear envelope that protects the host’s genetic material breaks down, making it easily accessible. HIV-1 is a type of retrovirus, called a lentivirus, that can infect non-dividing cells.
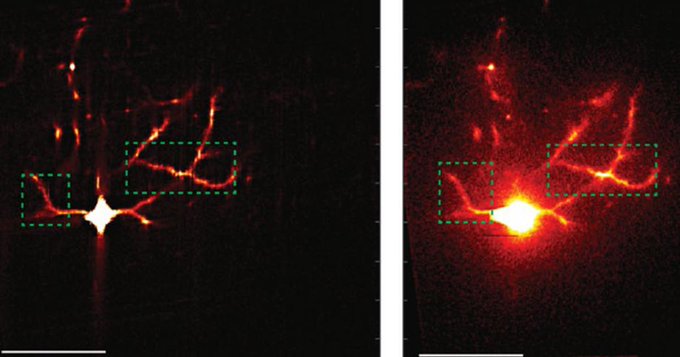
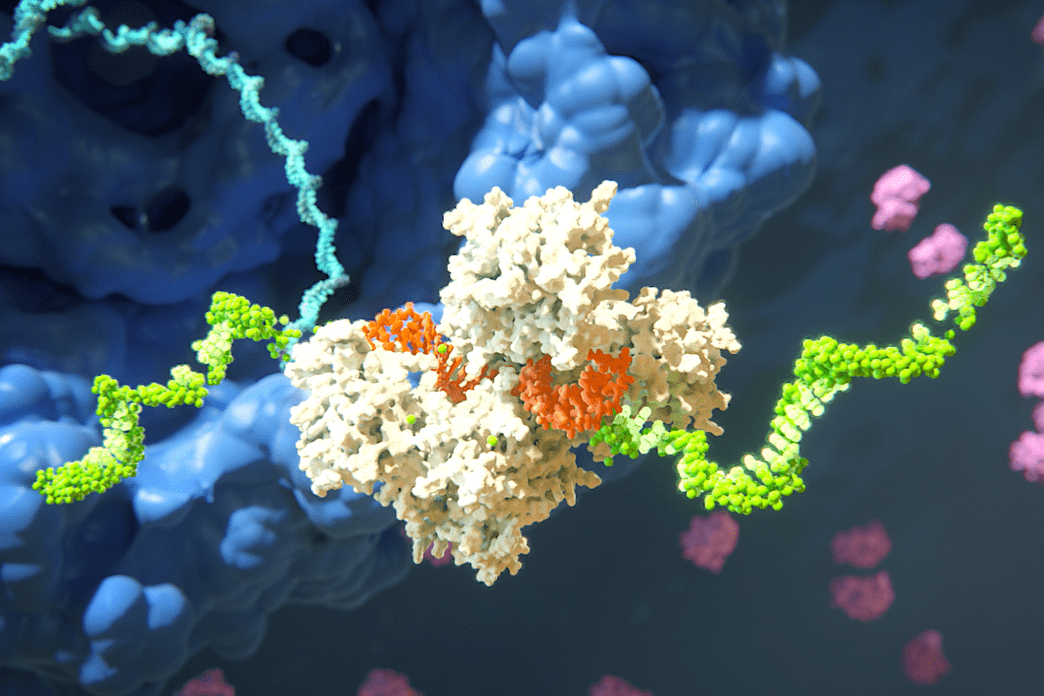
HIV-1 delivers its genome into the nucleus by packaging it into a large, cone-shaped structure called a capsid — but the exact mechanism has remained elusive for decades. Travel through the nuclear envelope occurs through, and is regulated by, nuclear pores, doughnut-shaped protein assemblies. Human cells have about 2,000 nuclear pores perforating the nuclear envelope. Some earlier evidence suggested that the capsid remains intact during its delivery into the nucleus — but this created a dimensional conundrum. The cone-shaped HIV-1 capsid is about 120 nanometers long and 60 nm wide — too large, researchers thought, to fit through the opening of the nuclear pore, measured at only 43 nm wide.
Members of the Schwartz Lab at MIT, in the Department of Biology, became interested in this question when a postdoc in the lab used cryo-electron tomography, slicing up sections of frozen cells to examine structures, to show that nuclear pores in the nuclear envelope are larger than 43nm. They deflate and shrink, it turns out, when removed from their native conditions. In native conditions, the nuclear pore complex is about 60nm wide — wide enough to accommodate the HIV-1 capsid.
Knowing that it could fit, a question remained: How can the capsid navigate the dense mesh of spaghetti-like proteins that act like a sieve in the nuclear pore channel? That spaghetti-like mesh allows small cargo to diffuse through, but prevents large cargo from entering unless it is escorted by proteins called nuclear transport receptors.
In an open-access paper published today in Nature, researchers present evidence that the HIV-1 capsid mimics the cell’s transport receptors to traverse the nuclear pore.
To support that conclusion, the researchers showed three things in vitro: that an HIV-1 capsid can deliver cargo through a nuclear pore analog; that the capsid can interact with the sieve of proteins in the nuclear pore channel; and that the capsid targets the nuclear pore in the absence of native transport proteins.
Nuclear transport receptors escort large cargo through the nuclear pore by “batting away” the spaghetti-like mesh of proteins inside the channel — like someone holding your hand and guiding you across a crowded dance floor. The HIV-1 capsid interacts with the spaghetti-like proteins, but its purpose is more like a Trojan horse — the capsid encapsulates the viral cargo, protecting it from detection in the cytoplasm and as it enters the nuclear pore complex.
“What’s really amazing about cells is that they are incredibly complex. What’s really difficult about studying cells is that they are incredibly complex,” jokes co-first author Erika Weiskopf, a graduate student in the Schwartz lab. “Biochemists are constantly trying to find ways to study their system in a simplified context, but still give it a flavor of cell biology.”
To do that, the Schwartz lab collaborated with Dirk Görlich, the director of cellular logistics at the Max Planck Institute for Multidisciplinary Sciences. Görlich is a co-senior author on the paper with MIT’s Boris Magasanik Professor of Biology Thomas Schwartz. Görlich’s lab has produced concentrated droplets of the spaghetti-like proteins found inside the nuclear pore, and those droplets allow and exclude cargo the same way a nuclear pore will. In experiments, fluorescently-labeled cargo did not enter the droplets, but fluorescently-labeled cargo packaged in an HIV-1 capsid was delivered. This indicated that the capsid could deliver cargo through a nuclear pore.
Using a biophysical binding assay, the researchers also showed that the HIV-1 capsid interacts with the proteins inside the channel. Different spaghetti-like proteins are found in different channel sections, such as at the cytoplasmic side’s entrance or only inside the channel; there are 10 such proteins in human cells. The capsid is a promiscuous binder — it can interact with all the spaghetti-like proteins found in the channel.
The capsid can target the nuclear pore complex even without the cell’s transport receptors, indicating that it is not commandeering native transport receptors to find and enter the nuclear pore. The team used a classic assay in the nucleocytoplasmic transport field to collect this evidence: When cells are treated with digitonin, their membranes become porous. Everything in the cytoplasm will leak out of the cells, but the nuclear envelope will remain intact. Despite the absence of native proteins, the capsid was attracted to the nuclear pore complex, a behavior indicative of a nuclear transport receptor.
Although the capsid behaves like a nuclear transport receptor to penetrate the nuclear pore, it is fundamentally different. A transport receptor doesn’t need to conceal material for delivery the way the capsid does to avoid detection.
These findings open new lines of inquiry for what the nuclear pore complex is capable of accommodating.
“The HIV-1 capsid is one of the largest things that we now know can go through the nuclear pore complex intact,” Weiskopf says. “It raises all kinds of questions — what other things could be going through the pore that we thought was impossible?”
Schwartz said another question is whether all of the 2,000 nuclear pores in human cells are identical or whether there is something that makes certain pores more amenable to allowing the capsid through.
The capsid is also known to be unusually elastic, a property that may be key for passage through the pore. Another interesting question for the field is whether the cone-shaped capsid gains entry into the pore by squeezing through.
Although the team has shown that the capsid can enter the pore, what happens at the other end of the channel is still unknown — whether the capsid fully or partially enters the nucleus or breaks down inside the channel. Weiskopf is working on perturbing parts of the capsid or the spaghetti-like proteins to learn more about which interactions are most important for successful capsid entry.
Although these results have expanded our understanding of the nuclear pore, much remains unknown, both for HIV-1 infection and for the transport process through the nuclear pore complex.
“The nuclear pore is such an important element of cell biology, we thought it would be interesting to understand it better — and that’s how we figured out that the pore is much bigger than we anticipated,” Schwartz says. “We will certainly try to see whether we can understand the mechanism of HIV-1 infection, how the capsid is released on the other side of the channel, and what factors are important there — and to what extent you can manipulate it or influence it for therapeutic applications.”