A new database of potential antibiotic targets
Raleigh McElvery
Many cells, including bacteria, are covered in a sugar-rich coating that protects their membrane and internal components. These sugars are often joined to other macromolecules, like proteins or lipids, to form glycoconjugates. The glycoconjugates that encrust bacteria help prevent them from “popping” under environmental stress, and facilitate host-pathogen interactions. Because the sugary layer perpetuates survival and virulence, researchers are looking for ways to create chinks in this microbial armor — or better yet, to prevent it from being made in the first place.
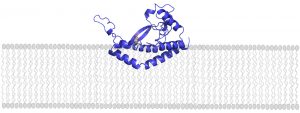
Glycoconjugates are built by many enzymes working in close succession at the cell membrane. One enzyme family, comprised of phosphoglycosyl transferases (PGTs), is responsible for catalyzing the first step in the assembly line. Of this large enzyme family, one subtype in particular stands out: “monotopic” PGTs, which are unique to bacteria and could serve as antibiotic targets. If researchers can develop drugs that inhibit monoPGTs, the sugar armor wouldn’t be built and noxious bacteria could be easier to defeat.
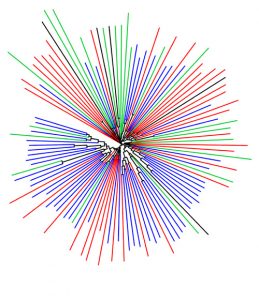
A new PNAS study co-authored by Professor of Biology and Chemistry, Barbara Imperiali, highlights the diversity and significance of these potential drug targets. Imperiali teamed up with graduate student Katherine O’Toole and Professor of Chemistry Karen Allen from Boston University to categorize over 38,000 different monoPGTs, compiling this information into the first database of its kind.
“We’ve taken an enzyme family that was once considered quirky and insignificant, and demonstrated that it’s actually very prevalent,” Imperiali says. “Hopefully the database will help us better understand these enzymes, their molecular pathways, and the human pathogens they support.”
Imperiali and her colleagues used sequence analysis of known monoPGTs to define a “signature” amino acid sequence. They leveraged this signature to identify the entire superfamily of monoPGTs amidst the 63,152 sequences downloaded from an online portal, which they then clustered into closely-related subtypes. The researchers also created a family tree, which included over 100 monoPGTs from diverse bacterial species. Imperiali hopes others will take advantage of this new information to pinpoint monoPGTs in pathogens of interest, and explore similarities and differences in related microbes and their enzymes.
The researchers’ analyses also revealed strange, new proteins that appeared to include two enzymes in one — a monoPGT fused to one of the other enzymes that typically play a separate role in the same sugar-modifying pathway. “It’s essentially one protein with two functions,” Imperiali explains. These fusion enzymes could reveal which enzymes “talk” to one another and work sequentially during the glycoconjugate-building process, she adds, revealing the complicated chain of events that creates the bacterial sugar shield.
The team even found cases where one monoPGT was fused to a member of a different PGT family — polytopic PGTs (polyPGTs). MonoPGTs and polyPGTs are involved in different pathways that each build glycoconjugates, so having a dual-function protein could allow cells to easily switch between mechanisms. Bacterial cells lack the organizational compartments that human and other eukaryotic cells have, so perhaps these fusion enzymes help exert control and order at different points in the cell cycle, Imperiali speculates. At the moment, though, the hybrid PGTs remain an evolutionary mystery.
While some researchers parse these ancient puzzles, others may use the database to inspire new drugs to combat antibiotic resistance. “At the end of the day,” Imperiali says, “we’ve shed light on a set of enzymes that could become pivotal therapeutic targets.”
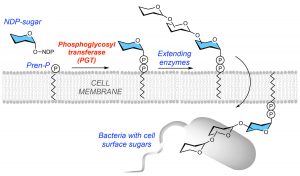
Glycoconjugates are built by many enzymes working in close succession at the cell membrane. PGTs catalyze the first step, and other enzymes continue adding sugars to build the protective layer that coats bacteria. All images are courtesy of the researchers.
Citation:
“Glycoconjugate pathway connections revealed by sequence similarity network analysis of the monotopic phosphoglycosyl transferases”
PNAS, online January 20, 2021, DOI: 10.1073/pnas.2018289118
Katherine H. O’Toole, Barbara Imperiali, and Karen N. Allen
